For Immune Cells, Discrimination is a Good Thing
Immune cells must be able to differentiate between good bacteria and bad bacteria to maximize the body’s chance at remaining healthy. A new study from scientists at Ludwig Maximilians Universitat (LMU) in Munich reveals how immune cells practice tolerance in response to good bacteria, even though everything their training has taught them pushes them to attack.
The training process teaches immune cells to discriminate between tissues of the human host - “self” cells - and proteins from a deleterious pathogen - “non-self.” Knowing the difference between the two of these entities prevents friendly fire but also ensures the immune system is prepared to launch an attack against a pathogen if necessary.
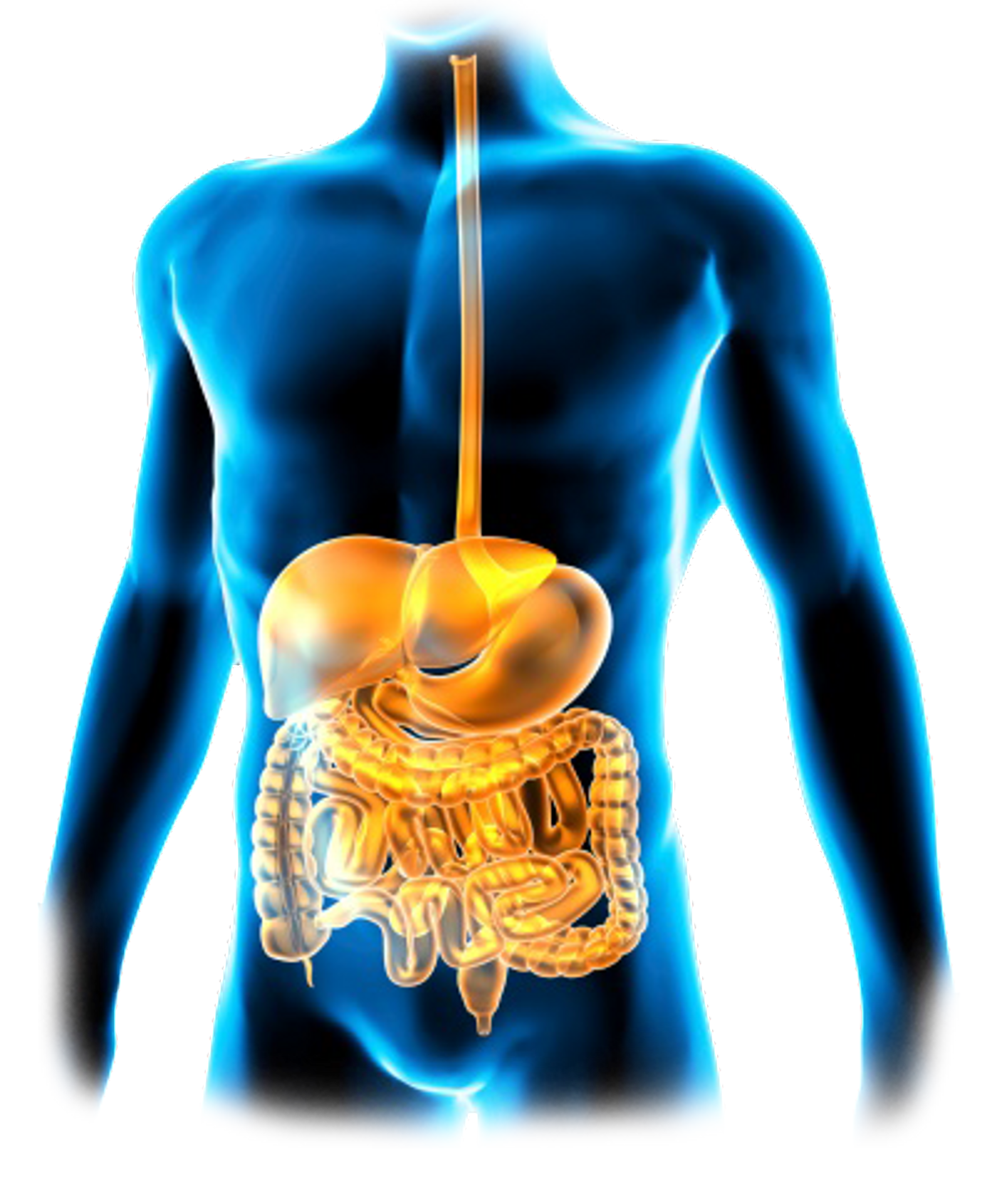
Despite receiving ample training, it is more difficult for immune cells in the epithelial lining of the gastrointestinal (GI) tract to make the distinction between self and non-self than it is for others because of their regular interactions with the microbiome, the “good” bacteria. Gut bacteria won’t be registered by the immune cells as “self,” but they still cannot be attacked without serious health repercussions.
The population of bacteria in the GI tract, called the “gut microbiome,” are part of a large community of beneficial bacteria in the body that influence health in numerous ways. A recent discovery from the new LMU study identified a mechanism that helps immune cells maintain a sort of “immunological equilibrium” in the GI tract, both protecting the good bacteria and defending against the bad bacteria.
The new mechanism clearly separates two opposite roles of immune players called dendritic cells (DCs). On one hand, the immune system counts on DCs to launch an immune attack in response to a pathogenic infection. On the other hand, DCs are also charged with promoting immunological tolerance, suppressing the immune response when needed.
DCs suppress the immune system by stimulating regulatory T cells (Tregs) that, as their name suggests, regulate the tolerance process and inhibit various inflammatory parts of the immune system. In the GI tract, epithelial DCs “recognize and internalize microbial proteins” before they travel to intestinal lymph nodes.
Once in the nodes, bacterial proteins are processed into small fragments which are displayed on the surface of the DCs as a way to flag down Tregs. Then, Tregs recognize the surface markers as a signal to begin suppressing any immune response against the specific markers held on the surface of the DCs, a reaction researchers believe to be unique to the particular marker being held on the cell surface.
This interactive behavior between DCs, Tregs, and microbial proteins from the GI tract is continuous, with Tregs and the immune system receiving a constant update of the goings-on in the gut microbiome, but a disruption can cause DCs to switch from suppressive behavior to stimulative behavior in terms of the immune response.
CD40, a signaling molecule and a surface receptor on DCs, sometimes acts as an alarm. When effector T cells bind CD40 on the surface of DCs, they transition into an immune-stimulatory state instead of the immune-suppressing state associated with Tregs.
LMU researchers demonstrated in mice models the DC transition, which depends on interactions between other immune cells and its surface markers. For example, when researchers triggered permanent CD40 signaling, severe colitis resulted. Colitis is an infection the GI tract characterized by inflammation.
In the case of CD40 signaling, DCs still make the trip from the GI tract epithelial tissue to the lymph nodes, but once they reach the nodes, they die before they can interact with Tregs, failing to initiate suppression of the immune system, resulting in colitis
"These findings show that interaction between dendritic cells and regulatory T-cells is essential for the maintenance of the correct immunological equilibrium or homeostasis in the gut," explained study leader Professor Thomas Brocker. The study was recently published in the journal Nature Communications.
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
SEP 03, 2024Microbiology Week Virtual Event Series 2024
- See More