A COVID-19 Vaccine Booster Now Recommended for Immunocompromised People
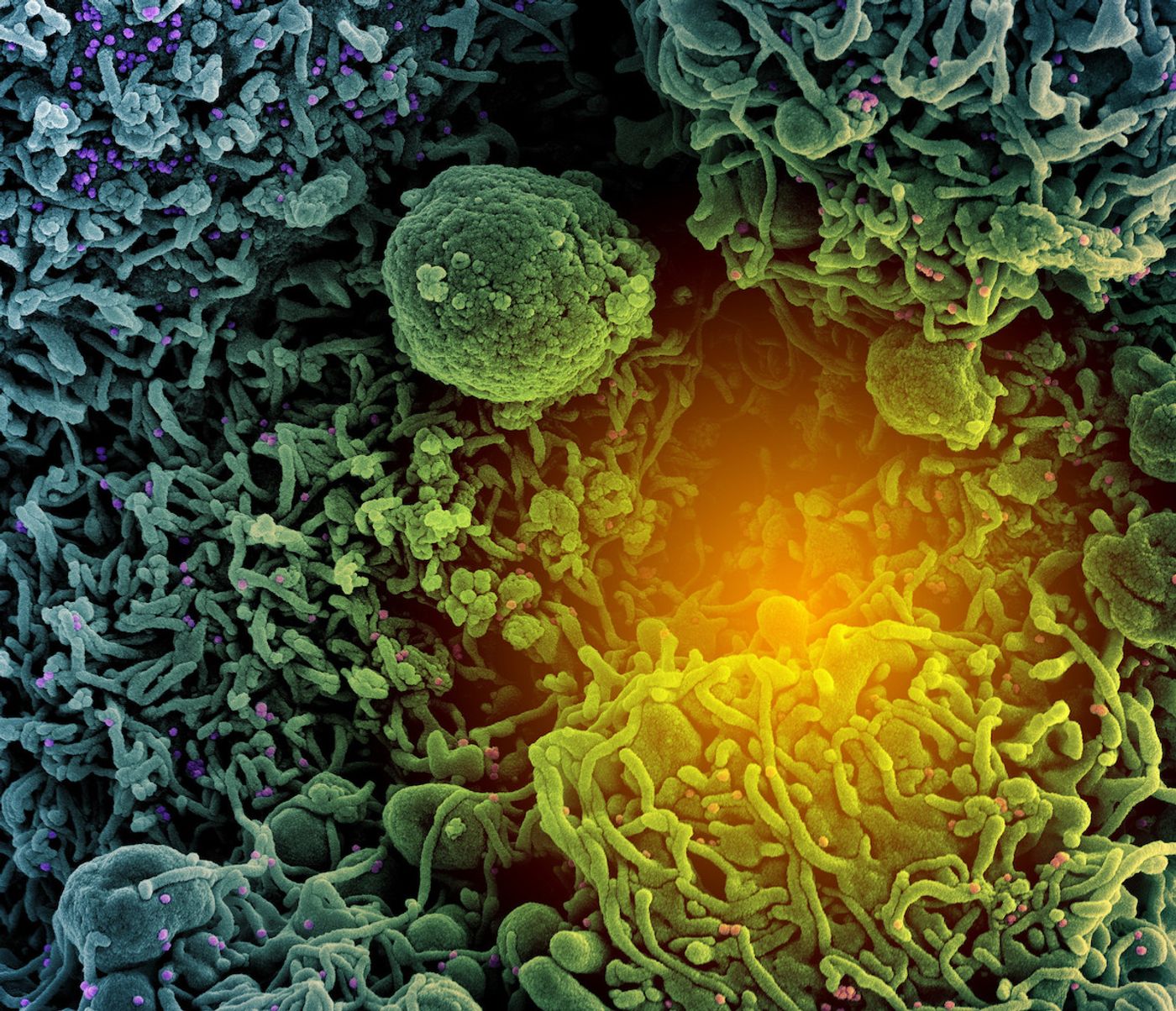
As Delta has become the dominant SARS-CoV-2 variant causing COVID-19 infections in the United States, researchers and health officials have been trying to determine whether the current vaccines can protect against this particularly contagious variant. Right now, the answer seems to be yes, the mRNA vaccines produced by Pfizer and Moderna are effective against the Delta variant, especially at preventing hospitalizations and deaths.
However, recent pre-printed work (not yet peer-reviewed) has suggested that the efficacy of these vaccines may decline as more unvaccinated people become infected and contagious. However, the work needs to be evaluated further before we can draw solid conclusions from it. Learn more about breakthrough infections from the video.
Related: COVID-19 Delta Variant Seems to Grow Faster & Be More Contagious
Officials are understandably eager to get as may people vaccinated as possible, for many reasons. For example, it's important to reduce the strain on our healthcare system, and the faster we can reduce the spread of the SARS-CoV-2 virus, the fewer chances it will have to mutate into other variants as well.
The Centers for Disease Control and Prevention (CDC), the Food and Drug Administration and other organizations are beginning to assess whether COVID-19 vaccine booster shots will be needed.
As of August 13, 2021, vaccine boosters have been authorized for a small number of people in select groups. According to CDC recommendations, the following individuals are eligible to receive an additional dose of the 2-dose vaccine they received. It's too soon to know whether an additional dose of the Johnson & Johnson (J&J) vaccine will provide any additional protection, so another J&J dose is not recommended at this time. Patients on the following list (copied directly from the CDC) who got a Pfizer vaccine should get another Pfizer dose if desired, and those who received a Moderna vaccine would get another Moderna dose if desired.
- "Active treatment for solid tumor and hematologic malignancies
- Receipt of solid-organ transplant and taking immunosuppressive therapy
- Receipt of CAR-T-cell or hematopoietic stem cell transplant (within 2 years of transplantation or taking immunosuppression therapy)
- Moderate or severe primary immunodeficiency (e.g., DiGeorge syndrome, Wiskott-Aldrich syndrome)
- Advanced or untreated HIV infection
- Active treatment with high-dose corticosteroids (i.e., ≥20mg prednisone or equivalent per day), alkylating agents, antimetabolites, transplant-related immunosuppressive drugs, cancer chemotherapeutic agents classified as severely immunosuppressive, tumor-necrosis (TNF) blockers, and other biologic agents that are immunosuppressive or immunomodulatory."
Right now, COVID-19 is surging in the United States again. About 129,000 new cases are being reported daily. It's estimated that 51 percent of Americans are fully vaccinated and about 60 percent have gotten one dose. Right now, approval to give the COVID-19 vaccines to children under age 12 is expected around mid-winter, though the United States is asking for approval to be fast-tracked. Results from trials are expected in the fall, and those results will have to be reviewed.
Researchers will be continuing to assess whether boosters will be needed for the elderly, or the general population. Unfortunately, the general public is unaccustomed to rapidly-changing and updated information regarding infectious disease, and keeping up with these shifts in knowledge has been challenging and confusing for many.
Source: CDC
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
SEP 03, 2024Microbiology Week Virtual Event Series 2024
- See More