Identifying New Drug Targets in Therapy-Resistant Cancers
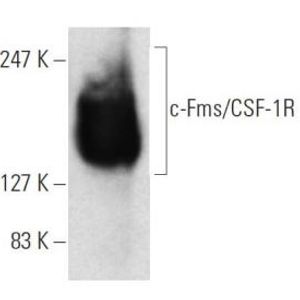
Recent research published in the Nature Partner journal Systems Biology and Applications examines how genome-wide data used in collaboration with systems biology analyses can potentially identify master regulators and new drug targets of therapy-resistant cancers.

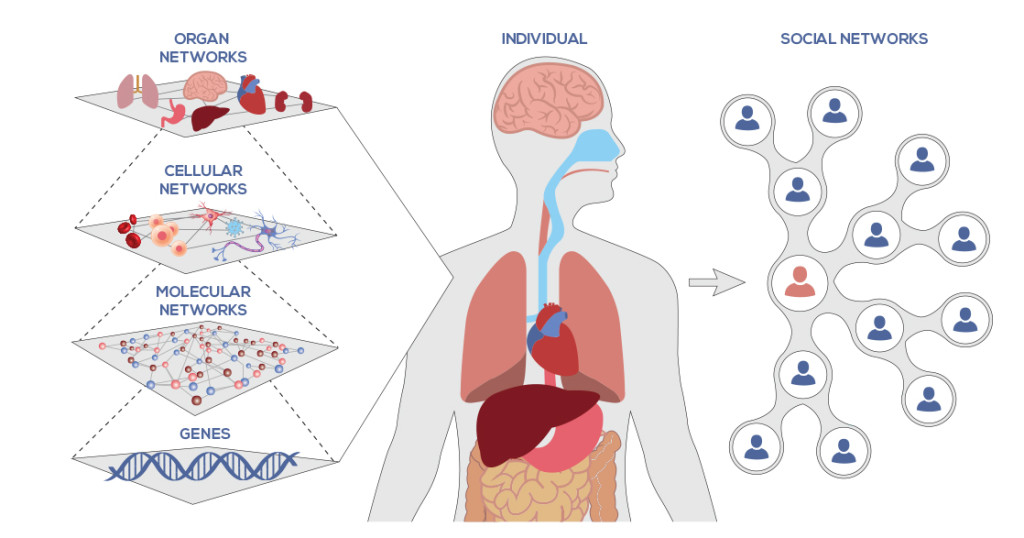
The findings explain how cancer takes hold and controls certain effector networks, a crucial area for the future of cancer therapy.
Cancer researchers are aware of the significance of epigenomic control in development and disease processes. To determine which cellular programs are silent and which are active, epigenomic signals beyond the genetic code of the DNA.
“Epigenomics does not directly affect the DNA itself,” Professor Fabian V. Filipp said. “A hidden layer of regulation controls the activity of genes. In cancer, if we disregard or insufficiently understand epigenomic networks, tumor cells have the ability to rapidly adapt to drug treatment and resistance may arise.”
To accomplish target-specificity, epigenetic master regulators in cancer allow target-specificity of their phenotypic program in cooperation with members of the transcriptional machinery. Therefore, the cancer systems biology research team at UC Merced developed a universal workflow for the elucidation of regulatory cooperation networks.
“By integrating different genomic features of complementary epigenomic and transcriptomic data, a highly specific and hyperconnected network can be identified,” explains Flipp.
Epigenomic target regions can define accessible bile chromatin and determine the target genes.
"The research portfolio of Professor Filipp builds an important bridge between clinical and computational sciences," Vice Chancellor for Research Samuel Traina said. "Professor Filipp runs an interdisciplinary research program that serves and positively impacts the educational needs of California's Central Valley".
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
SEP 03, 2024Microbiology Week Virtual Event Series 2024
- See More