Potential AML Drug Encourages Cancer Cell Mapping
International researchers have sought to be the first in mapping family trees of cancer cells in acute myeloid leukemia (AML) to help them understand how this particular blood cancer responds to a new drug by the name of enasidenib.
The most common and aggressive blood cancer in adults, AML is incurable in most patients. Roughly 12-15% of individuals diagnosed with AML have a mutation in the IDH2 gene which inhibits bone marrow cells from differentiating, or maturing, into blood cells that are required for life. Instead, these immature cells accumulate in the bone marrow and blood, a hallmark of AML.
"Enasidenib is an important new treatment. However, the initial studies did not show which AML cells responded to enasidenib and started to differentiate again. It was also unclear how the cells become resistant to therapy," said Dr. Stéphane de Botton, a physician in the hematology department at Gustave Roussy. "We wanted to answer these questions."

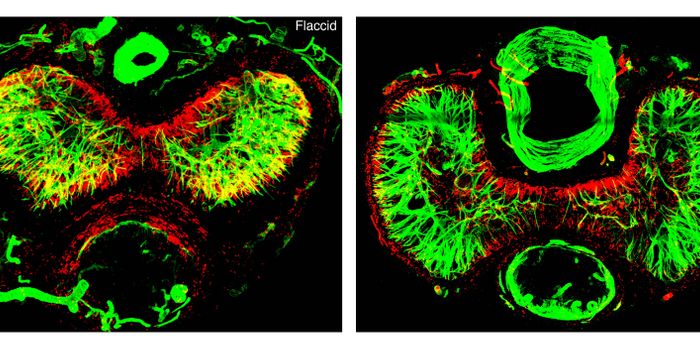
Samples that were taken from 37 AML patients who responded to enasidenib in the clinical trial were investigated by researchers who examined biomarkers on the surface of the bone marrow cells to identify the different populations of bone marrow cells, from the immature, undifferentiated cells, called progenitor cells, through to mature, differentiated cells.
"You can imagine the bone marrow as an assembly line that constantly needs to produce mature blood cells," said co-author, Dr. Lynn Quek, MRC clinician scientist and consultant hematologist at the MRC Weatherall Institute of Molecular Medicine. "In a patient before treatment, this assembly line becomes blocked. Using specialist techniques, we studied bone marrow cells from patients to find out how the bone marrow assembly line becomes blocked and how enasidenib helps to promote differentiation to unblock the assembly line."
AML is a result of errors in DNA sequence, or mutations, in blood cells, the team studied the genetic make-up of AML cells. Results showed that AML cells retrieved from the same patient can be categorized families that share the same genetic mutations, called clones. The cells that belong to the same clone or family are a result of the same ancestor cell. Understanding the relationship between clones is crucial to providing information on the origins and development of AML.
"When an AML patient has a bone marrow test, we are taking a snapshot of the family tree of leukemia cells," said Dr. Quek. "As we treat the AML, there are shifts in the family dynamics as some clones will die and others will grow. In every cancer, there are several families or clones of cancer cells. In AML we were able to see how these responded to enasidenib. We used techniques to study genetic mutations on a cell-by-cell basis and re-constructed the family tree of a patient's AML. We then tracked changes in the family of AML cells as they responded to enasidenib and as patients lost response to the drug. This is the first time that anyone has done such a detailed study at a single cell level. As enasidenib is a new anti-leukemic drug, it was vital to understand the effects of the drug on leukemic cells."
-
MAY 07, 2024Is It Anti-RNP or Anti-Sm/RNP?
- See More
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
-
MAY 23, 2024For the Love of Digital PCR 2024
- See More