Drug Combination May Be Effective for Melanoma
Protein kinase inhibitors have been noted to be the most effective class of drugs in the treatment of melanoma—a deadly skin cancer of the melanocytes.
However, these inhibitors, in many cases, result in drug resistance causing a relapse in the patient.
Now, a new study published in Molecular Cancer Therapeutics suggested that the combination of kinase inhibitors with experimental drugs called ‘ribonucleases’ may be able to combat drug resistance and leading to better results. "We discovered that this ribonuclease drug could be paired favorably with other cancer chemotherapeutic agents, and not only that, the pairing made logical sense in terms of the underlying biochemistry," says Ronald Raines, the Firmenich Professor of Chemistry at MIT and senior author of the study.
Ribonucleases are enzymes that break down RNA molecules. They are produced by all human cells that and work to degrade cellular RNA and help defend against viral RNA. Overall, ribonucleases hold a unique ability to kill cells by damaging their RNA. "That means that should ribonuclease invade cells, there is an unbelievable defense system," Raines says.
In particular, the study observed an unexpected link between ribonucleases and enzymes--the protein kinases (targets of protein kinase inhibitors). This led researcher’s to the discovery of two drugs that can kill cancer cells at high efficacy. The discovery was the result of researchers using a ribonuclease inhibitor protein in human cells instead of E. coli, which they normally use to produce the protein. They found that the human-cell-produced version bound to ribonucleases 100 times more strongly.
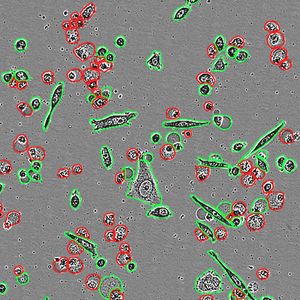
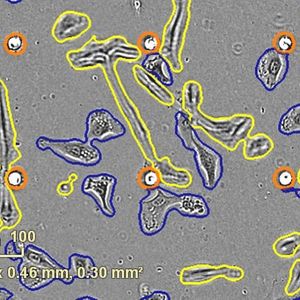
MIT researchers are developing ribonucleases as potential drugs to treat cancer. In a new study, they found that the drugs work better when given with already-approved drugs known as kinase inhibitors.- MIT News
Image Credit: MIT News
Encouraged, the researchers created a ribonuclease drug that has been modified so ribonuclease inhibitors don't bind as tightly—one version of such drug has already made it to phase 1 clinical trial, stabilizing the disease in about 20 percent of patients.
Previously, researchers thought that human cells were somehow modifying the inhibitor to make it bind more tightly. These assumptions were confirmed when studies revealed that the inhibitor had attached phosphate groups which meant that it was phosphorylated. The "phosphorylation" allowed the inhibitor to bind strongly than previously suspected.
Source: MIT, Molecular Cancer Therapeutics
-
-
APR 30, 2024Immuno-Oncology Virtual Event Series 2024
-
MAY 07, 20243rd International Biosecurity Virtual Symposium
- See More