This year, doctors will diagnose over 240,000 new cases of breast cancer in women. Mammograms have been the standard first line of defense in breast cancer detection, but not every lump points to cancerous origins. Getting the right diagnosis is crucial to prevent unnecessary treatments, and a new pill that lights ups breast cancer cells could help doctors better make this call.
“There’s a lot of controversy right now about when patients should start screening for breast cancer,” said Greg Thurber, senior investigator. “Screening can potentially catch the disease early in some patients, but false positives can lead to unnecessary, aggressive treatments in patients who don’t need them. We don’t know how to select the right patients to treat. Our work could help change that.”
At the 251st National Meeting & Exposition of the American Chemical Society (ACS), researchers from the University of Michigan announced a pill that selectively fluoresces breast cancer tissues in mice. It works by binding to cancer cells or blood vessels unique to breast tumors. Then, under near-infrared light the pill causes cancer tissues to fluoresce, taking the guesswork out of determining whether a lump is malignant or actually benign.
In a standard mammography screen, X-ray radiation reveals the presence, location, and size of lumps in breast tissues that otherwise can’t be detected by touch alone. Even so, one drawback of mammography is it can lead to breast cancer false-positives, as the presence of a lump can’t indicate cancer with certainty. Often, doctors have to further validate mammography findings via invasive biopsies. The light-up pill could change how mammography results are read, and thus relieve some patients of needless procedures.
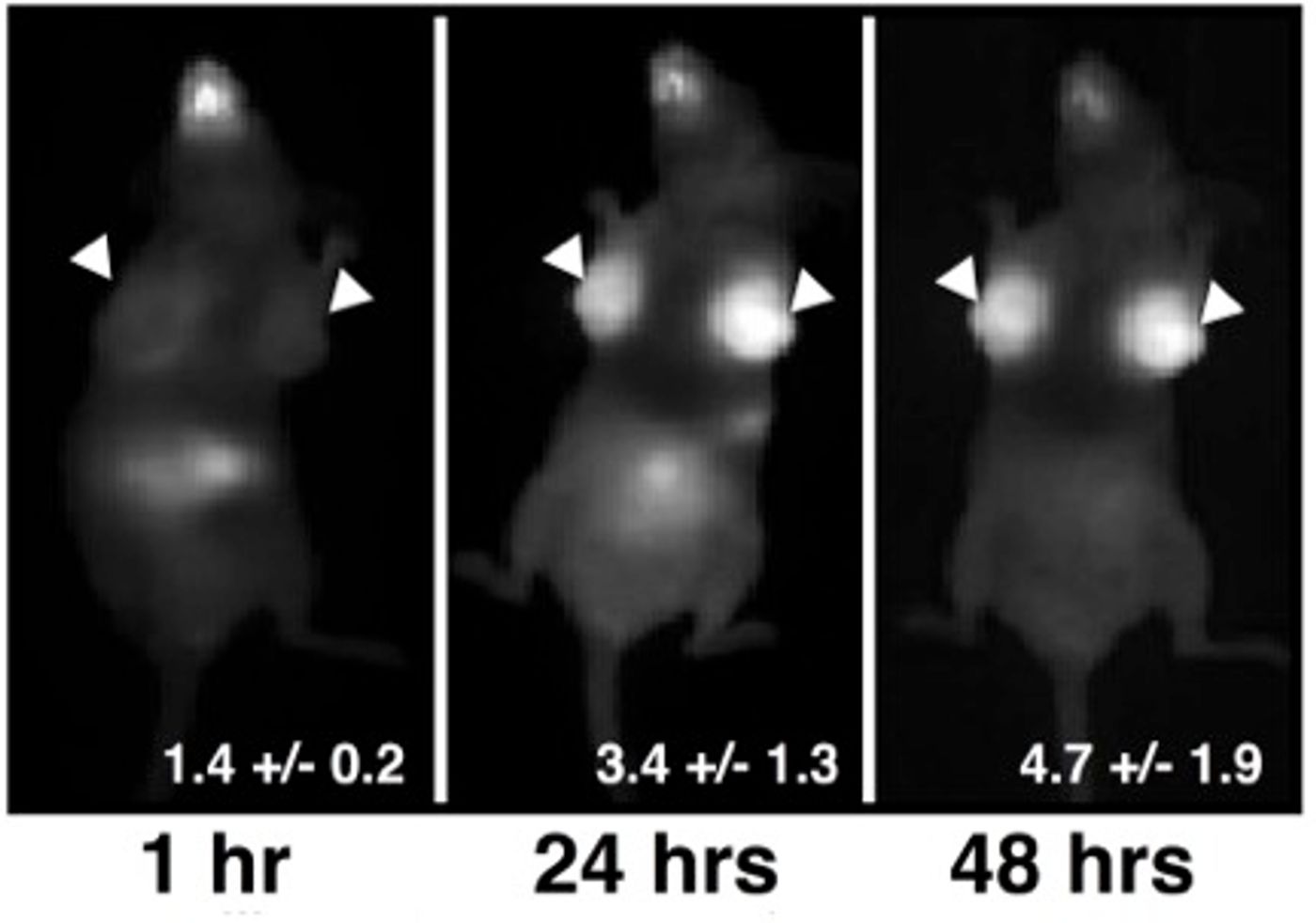
In a mouse model of breast tumor, the team found that the pill significantly absorbed into the bloodstream – at 50-60 percent – and that it selectively highlighted breast cancer tissues. In contrast, there was little activity in the surrounding background tissues, indicating a high binding specificity.
For the next phase, Thurber and his team are working to modify the formula, making it suitable for human clinical trials. Additionally, the team aims to enhance detection of not only breast cancer, but to differentiate between different sub-types of breast cancers with the pill. Knowing which types are slow-growing or invasive could allow doctors to further tailor therapies for their patients. While clinical trials could be a long way in the future, Thurber noted that they dye is already in use in Europe.
Additional source:
ACS press release
 In a mouse model of breast tumor, the team found that the pill significantly absorbed into the bloodstream – at 50-60 percent – and that it selectively highlighted breast cancer tissues. In contrast, there was little activity in the surrounding background tissues, indicating a high binding specificity.
In a mouse model of breast tumor, the team found that the pill significantly absorbed into the bloodstream – at 50-60 percent – and that it selectively highlighted breast cancer tissues. In contrast, there was little activity in the surrounding background tissues, indicating a high binding specificity.







