When it comes to some cancer types, more testing may not always pay off. It may, in fact, waste millions of dollars that could go towards other healthcare burdens. That’s what Johns Hopkins doctors report, after finding that for a common type of noninvasive breast cancer, routine testing at the onset of diagnosis may be
redundant and highly wasteful. How wasteful? The scientists estimated at least $35 million could be saved annually if this initial testing was not made so routine.
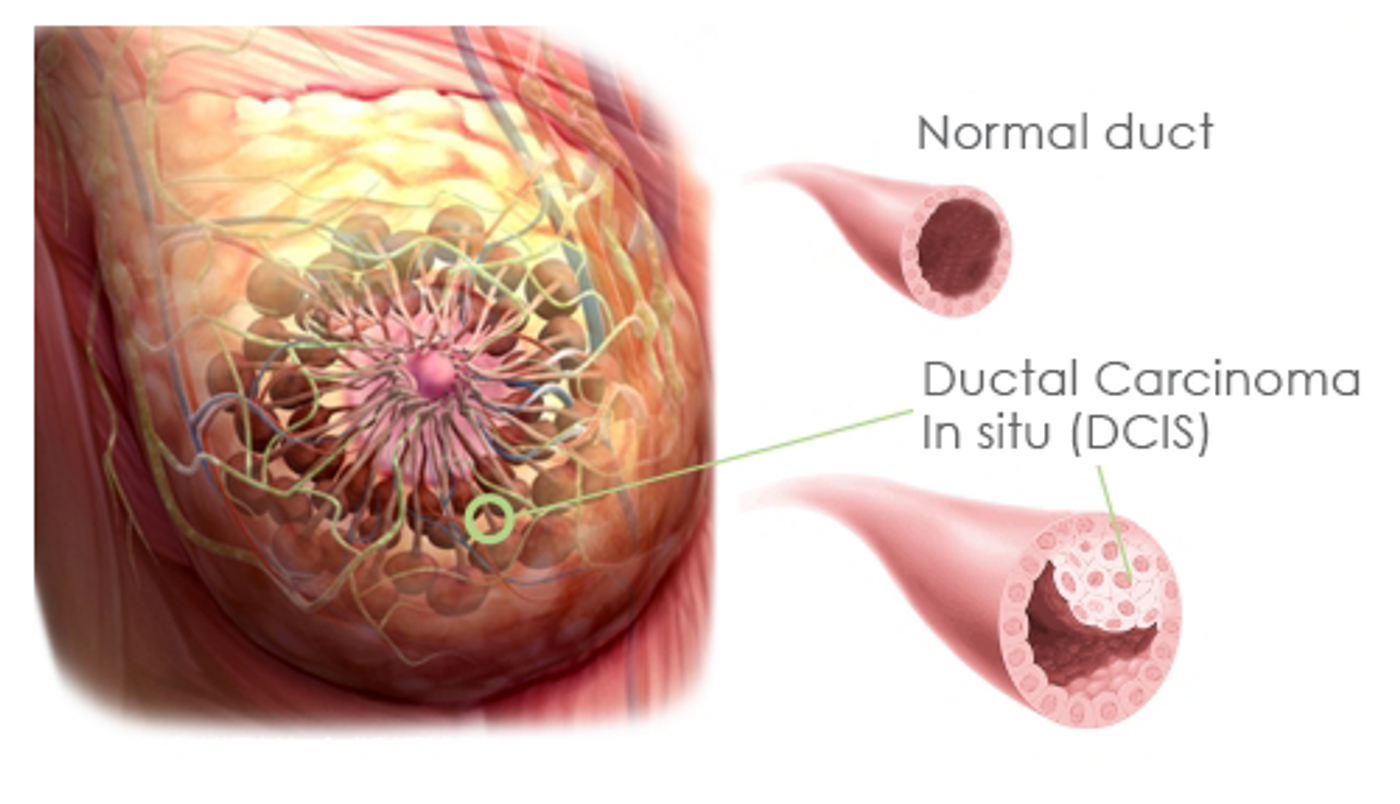
Every year, an estimated 60,000 patients in the US are diagnosed with a noninvasive breast cancer type known as ductal carcinoma in situ (DCIS). This condition is marked by presence of cancer cells in the milk ducts or lobules, which have not spread to anywhere else.
According to Hopkins researchers, it’s become almost a reflex for doctors nationwide to order core needle biopsies for hormone receptor testing following a DCIS finding. The routine procedure determines whether the cancer is noninvasive or invasive, and tests for the presence of estrogen and progesterone receptors.
But, regardless of the outcome of the biopsy, doctors end up making the same recommendation: lumpectomy or mastectomy to remove the cancerous tissues. This is the first line of argument against the needle biopsy.
Furthermore, the first round of testing shows itself to be essentially redudant later on. Pedram Argani, senior lead of the study said, “Almost 20 percent of the time, the excision reveals that the noninvasive DCIS that the core needle biopsy caught is actually accompanied by invasive breast cancer. So you have to receptor-test the invasive cancer that the core biopsy missed. That means the testing of the core biopsy with DCIS was useless."
But perhaps the most compelling argument to drop the initial needle biopsy may be its demonstrated lack of efficacy on patient survival. The premise behind this treatment is to catch hormone receptor-positive DCIS types, for which tamoxifen can be taken to inhibit the receptors and prevent cancer recurrence. However, the effect of this treatment overall is modest at best, while the side effects can be severe.
"Two-thirds of our DCIS study patients did not receive hormone therapy," said Argani, "even after excision confirmed their hormone-receptor positive DCIS status. So analyzing their needle biopsies or surgical excisions for receptors would have clearly been unnecessary for them."
What it amounts to, in the researchers’ perspective, is an expensive medical bill that’s not necessary. Every early receptor test costs about $140 per patient. "Extrapolating that to the roughly 60,000 U.S. patients diagnosed with DCIS yearly puts the cost at about $8.5 million," added Argani. "The unnecessary cost of automatically testing DCIS in excisions when the information is not used is $26.5 million, which adds up to $35 million dollars per year in the U.S."
So persuasive are the results that the Johns Hopkins Hospital have already abandoned this practice at the start of the year, 8 months before the formal publication of the study. Perhaps other hospitals will also follow suit.
Additional source:
Johns Hopkins press release
 Every year, an estimated 60,000 patients in the US are diagnosed with a noninvasive breast cancer type known as ductal carcinoma in situ (DCIS). This condition is marked by presence of cancer cells in the milk ducts or lobules, which have not spread to anywhere else.
Every year, an estimated 60,000 patients in the US are diagnosed with a noninvasive breast cancer type known as ductal carcinoma in situ (DCIS). This condition is marked by presence of cancer cells in the milk ducts or lobules, which have not spread to anywhere else.







