The Cues that Tell Pancreatic Cancer to Steal
Acting like cunning thieves in the night, pancreatic cancer can hack into their microenvironment to selfishly steal the essential nutrients for its own survival. A new study highlights the mechanisms behind this process, and points to a promising drug candidate that may prevent theft of the nutrients.
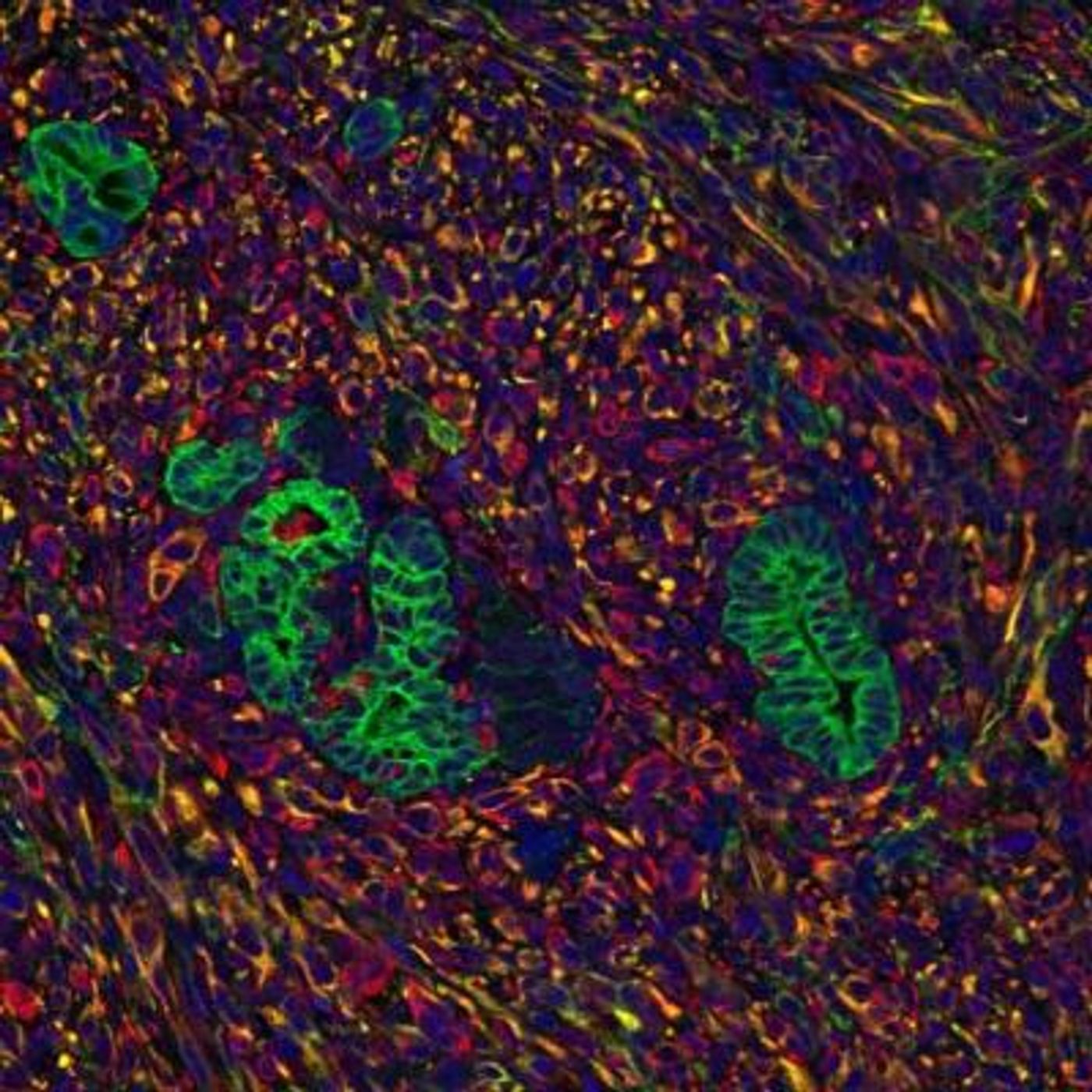
Indeed, scientists have shown that the stromal microenvironment, made up of fat and connective cells that provide structural support for tissues and organs, plays a big role in supporting tumor growth. But exactly how this happens remained a complex mystery.
To understand how tumors hack into this microenvironment, researchers at the Salk Institute replicated this biological process with and without signaling chemicals from the pancreatic stroma. "We worked out a culture system so that we could grow human pancreatic cells in a three-dimensional system in both the presence and absense of stromal signals," said Mara Sherman, the study’s first author.
The team showed that tumors relied on stromal molecules to mobilize their entire metabolic process. In essence, signaling molecules trigger the tumors to accelerate their growth and overall metabolism. At the epigenetic level, the tumors had increased histone acetylation at transcriptionally enhanced genes, which suggests a deeper level of genetic alteration.
"The tumor is essentially hacking into that stromal microenvironment and grabbing what it needs to up its metabolism," said Michael Downes, one of the study’s co-authors.
The Salk team hypothesized that changes at the epigenetic level implicates the pancreatic cancer epigenome as a therapeutic target. That is, blocking the signals at the epigenetic level may prevent the tumor cells from hacking into its microenvironment.
To test this hypothesis, the team administered an epigenetic inhibitor known as JQ1 into their cancer models. In cells, the drug blocked the stromal signals and reversed the genetic changes associated with increased metabolism. In a mouse model, the drug slowed tumor progression significantly.
The team plans to follow-up this pathway with JQ1 and other inhibitors to understand whether it will be effective in humans. Of note, pancreatic cancer is notorious for its lethality. Only about 25 percent of patients survive past the one-year mark from diagnosis. Thus, any insight that can be translated into drug therapies offers huge hope for patients.
Additional sources: Salk Institute