FDA Approves First Gene Therapy for Leukemia
For patients battling leukemia, there is hope in the form of a newly approved gene therapy. Called Kymriah, this is the first gene therapy that the FDA has ever cleared for the treatment of a cancer.
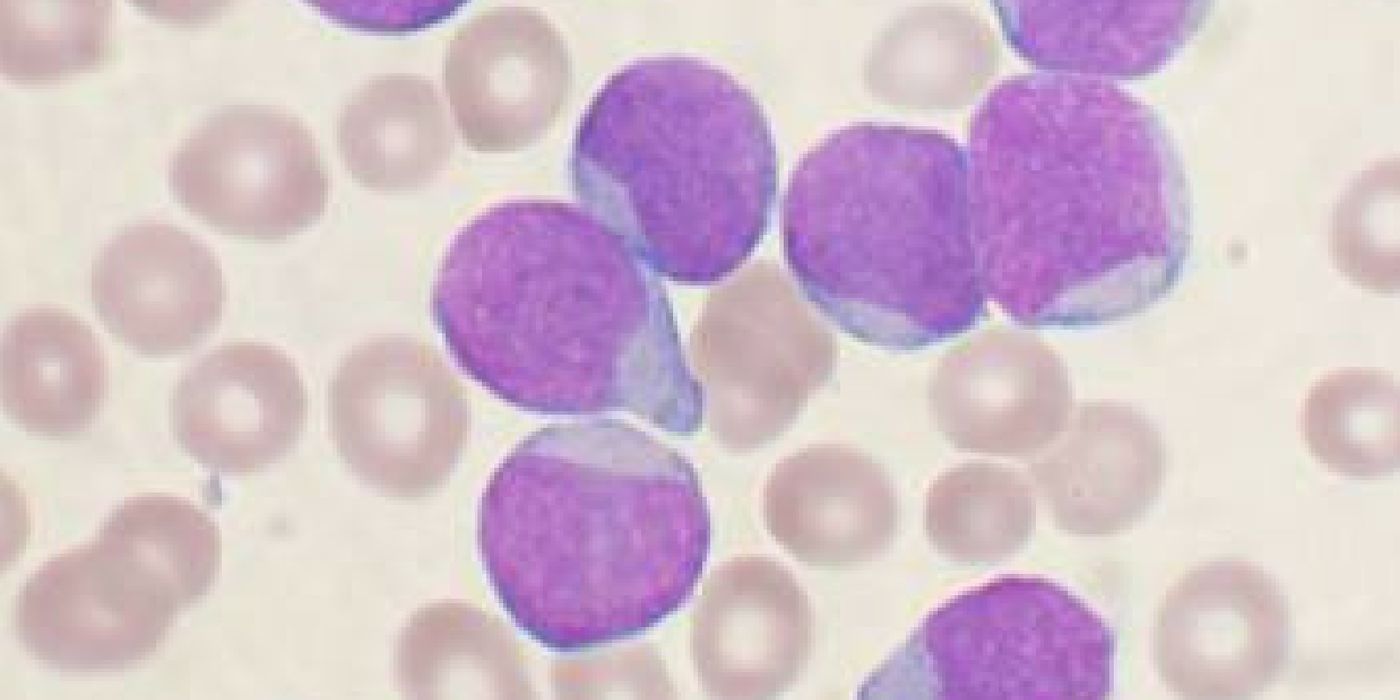
The new treatment is unlike any of the current leukemia treatments on the market. Instead of a toxic cocktail of drugs, each dose of the Kymriah is made up of the patient’s own immune cells. The premise of Kymriah rests on chimeric antigen receptor T-cell therapy (CAR-T), whereby patient’s T cells are genetically modified to be better at recognizing and killing the leukemia cells. In essence, a patient’s immune cells get outfitted with new armor against the cancer.
"We're entering a new frontier in medical innovation with the ability to reprogram a patient's own cells to attack a deadly cancer," said Dr. Scott Gottlieb, the FDA Commissioner.
This type of immunotherapy technique is also being tested for a variety of other conditions. However, Kymriah, manufactured by Novartis, nabbed the first historic approval from the FDA. The drug is approved for patients up to 25 years old battling acute lymphoblastic leukemia (ALL) that hasn’t responded to other drugs.
In the most recent clinical trial, researchers gave Kymriah to leukemia patients who had exhausted other first-line treatments. Kymriah allowed 83 percent of patients to live cancer-free for at least three months. It also gave patients an 89 percent chance of surviving at least 6 months, and a 79 percent chance of surviving past the one year mark. Oncologists call this nothing short of miraculous given the aggressive nature of the leukemia in patients who had limited alternatives.
“Kymriah is a first-of-its-kind treatment approach that fills an important unmet need for children and young adults with this serious disease,” said Peter Marks, who directs the FDA’s Center for Biologics Evaluation and Research (CBER). “Not only does Kymriah provide these patients with a new treatment option where very limited options existed, but a treatment option that has shown promising remission and survival rates in clinical trials.”
Though considered a rare disease, acute lymphoblastic leukemia (ALL) is the most common form of childhood cancer, peaking in early childhood between the ages of 2 and 4. The cancer involves the overproduction of immature white blood cells in the bone marrow. These cancerous cells invade the blood and other organ systems, causing massive cell death and damage in a short amount of time. Radiation therapy, bone marrow transplantation, and chemotherapy agents like methotrexate, vincristine, and cytarabine, are effective against ALL. However, it doesn’t work for everyone, and remission isn’t a sustained outcome in 15 to 20 percent of children in whom the cancer makes a dreaded comeback.
Unlike a conventional cancer drug, the CAR T-cell therapy is more potent because it can be self-sustaining. That is, even though it’s a one-time therapy, once re-introduced into the patient’s body, the engineered T-cells are expected to multiple and form an army against the cancer cells. This means that patients should be continually protected by the ‘living drug,’ much in the same way that vaccines protect us against pathogens.
The new treatment comes with a hefty price tag of $475,000. Novartis indicated they were trying to strike a balance between allowing patients to access the treatment while also allowing the company to recoup its investments. And though it sounds enormously high, some experts had put the estimate of one Kymriah dose at between $700,000 to $750,000.
Interestingly, Kymriah comes with a money-back guarantee -- patients who do not respond to Kymriah within a few months won’t be charged, Novartis said. "Novartis is collaborating with (Centers for Medicaid Services) to make an outcomes-based approach available to allow for payment only when pediatric and young adult ALL patients respond to Kymriah by the end of the first month. Future potential indications would be reviewed for the most relevant outcomes-based approach," the drug company said in a statement.
Because Kymriah is not a standard drug by any definition, patients have to travel to a select 32 sites around the country for the treatment. There, the patient’s cells will be harvested and shipped off to a Novartis lab in New Jersey for modification. Roughly three weeks later, the genetically engineered cells will be injected back into the patient to fight the leukemia cells.
As with any medical treatment, Kymriah treatment can come with some adverse events. Because the treatment heavily involves the immune system, patients may be at risk for cytokine release syndrome (CRS), which is an immune response to the newly modified T-cells. Patients may get fevers, flu-like symptoms, and neurological events. Patients are also at higher risk for infections because the CAR T-cells could also kill the normal B cells that produce protective antibodies.