Gastric Cancer Screening Tool Using Serum Biomarkers Being Studied in South Korea
A new study focused on Gastric Cancer (GC) is evaluating a diagnostic screening tool utilizing a serum biomarker test panel which includes pepsinogen, H. pylori IgG, and Trefoil factor 3 (TFF3). An additional outcome is to investigate the ideal timeframe intervals for performing GC screening endoscopies in South Korea.
The observational study is still recruiting patients and aims to have their analysis completed by 2022. The study is designed to survey newly diagnosed GC patients and a healthy control group. There are multiple centers involved in the study and recruitment of patients. As reported by the researchers in the clinical trial documentation, Korea has the highest incidence of GC; their focus is to help determine factors that will provide early diagnoses which have a more favorable prognosis. There are multiple factors that increase the risk of gastric cancer including age, gender, genetics, family history, race, obesity, smoking, and H. pylori infection history.
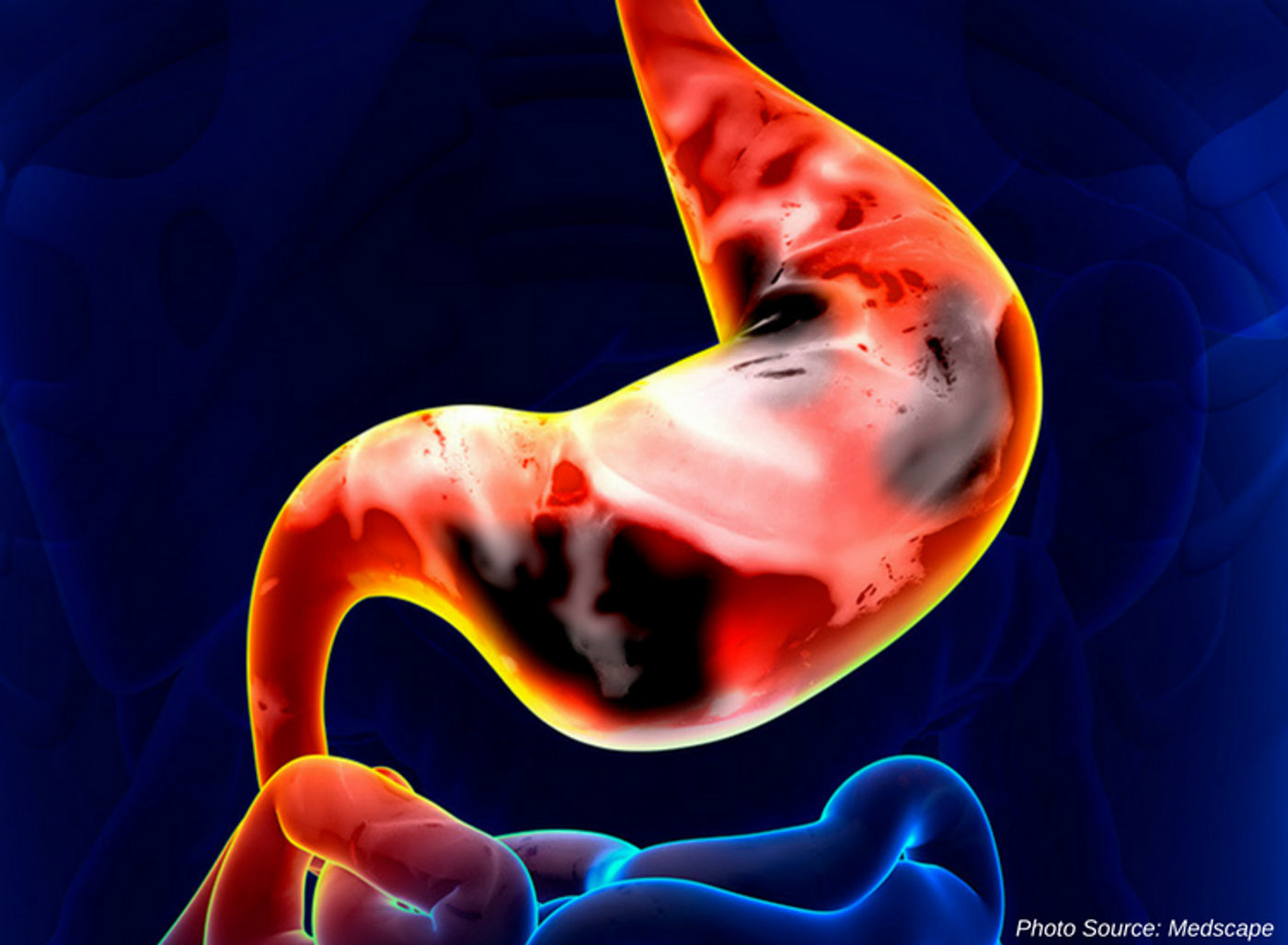
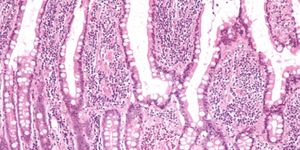
There is an acknowledged precancerous cascade of changes resulting in GC. GC traditionally begins with intestinal metaplasia which is recognized as a precancerous change. The mucosa of the stomach lining yields initially to inflammation and is followed by damaging intestinal metaplasia (usually found as result of Helicobacter pylori infection). Normal gastric mucosa is replaced by intestinal cells with partial or complete loss of the molecular features of gastric cells in the lesion location.
The search for accurate, less invasive and expensive testing is an important and valid endeavor for all conditions, especially for cancer. Current testing and screening includes serum pepsinogen (PG 1, PG II, and PG I/II ratio) which is reported to be a marker of mucosal atrophy; low values have consistently been found in GC patients. Mucosal atrophy is often a strong indicator that a precancerous state is developing. However, there are other possibilities for reduced levels of PG I or PG II. This study looks to add TFF3 testing; TFF3 is a secretory protein expressed in gastrointestinal mucosa. Recently published literature found with 73.4% sensitivity and 92% specificity that the optimal cutoff value of serum TFF3 was >8.9 ng/mL for GC diagnosis. Another group recommended a TFF3 cutoff value of 3.6 ng/mL with 80.9% sensitivity and 81% specificity. Of note, herein lies a challenge of determining appropriate reference ranges or cutoffs for diagnosis. TFF3 also identified diffuse-type GC at 68.0% and 92.0% respectively. TFF3 is potentially a more stable marker than the serum PG test which had a 69.6% sensitivity and 68% specificity. H. pylori serology, while not specifically outlined in the trial information, can be stratified by the degree of mucosal atrophy. Reportedly the population in East Asia, Eastern Europe and parts of Central and South America have high seropositive H. pylori antibody rates so using this alone as a screening tool will have very low specificity for GC or precancerous change detection. Including PG levels as a predictor combined with H. pylori seropositivity increases that specificity. Combining pepsinogen, H. pylori IgG, and TFF3 values into a new diagnostic guide together could improve gastric cancer screening.
Sources: NIH US. National Library of Medicine, Clinical and Experimental Medicine, Gut Liver Journal, Gastroenterology, Gastroenterology and Hepatology Journal, Disease Markers,