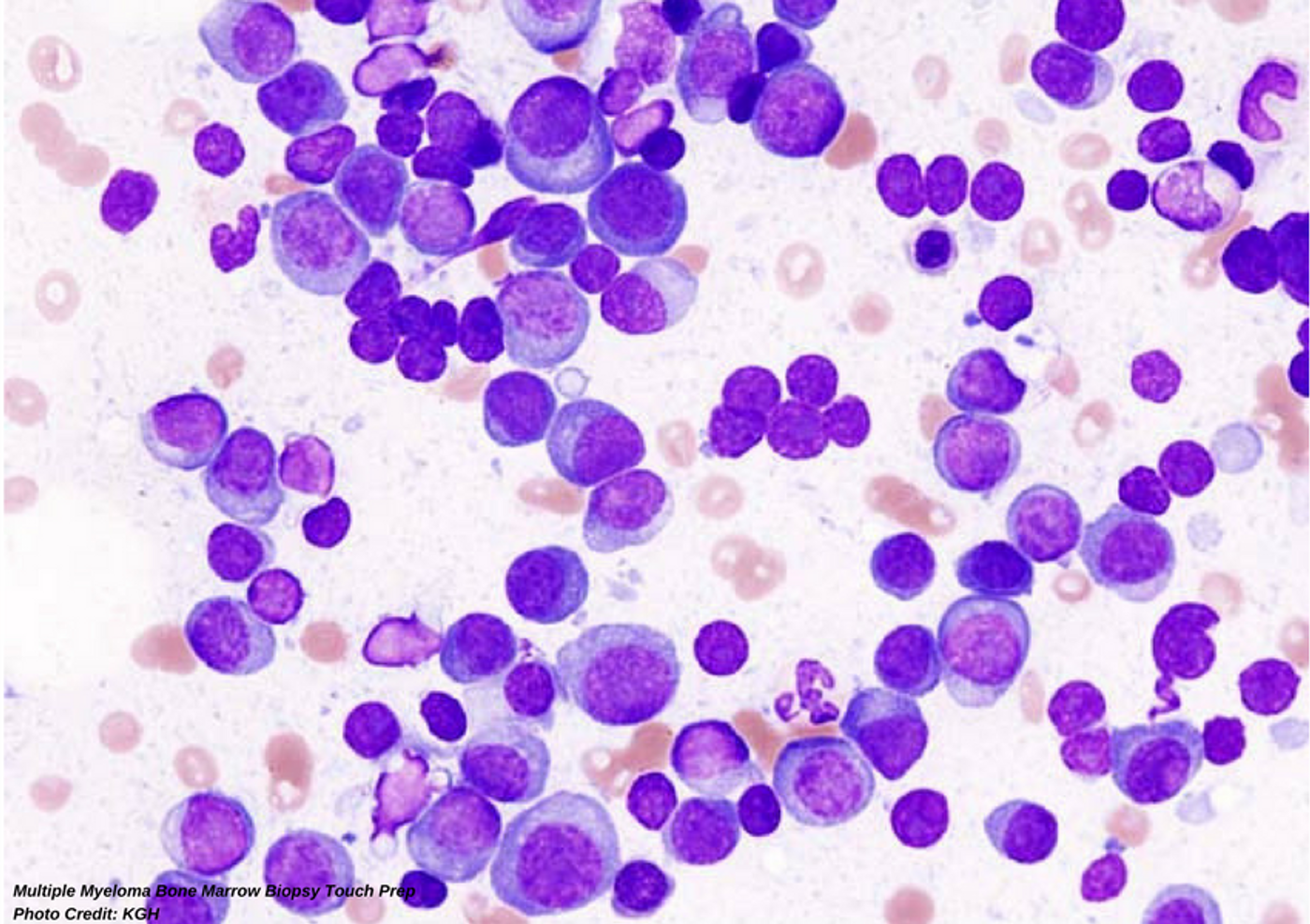
The Microfluidic Device: Future Replacement for Bone Marrow Biopsy in Multiple Myeloma
Collaborators from multiple research institutions shared their findings on a new diagnostic device in the journal Integrative Biology in late April 2018; the group developed and tested a less invasive diagnostic and monitoring tool for Multiple Myeloma (MM). This disease occurs in the bone marrow and currently requires a bone marrow biopsy for diagnosis and subsequent follow ups to monitor disease. Bone Marrow biopsies are incredibly painful and, while incredibly rich in diagnostic value, they are terrible for patients to endure. This research promises a new option where a plastic chip-based technology is used with whole blood to detect MM cells or circulating plasma cells (CPCs).
The chip is termed a microfluidic device with monoclonal anti-CD138 antibodies. The researchers tested the chip with whole blood drawn from patients with a known immuno-phenotype (CD38/CD56/CD45 panel positive) for Monoclonal Gammopathy of Unknown Significance (MGUS), Smoldering MM (SMM), and symptomatic MM. Researchers found that 78% of patients with MGUS were correctly identified and 100% of all MM/SMM patients were identified. Their positive and negative controls were also noted to be accurately identified. Not only was presence of CPCs detected accurately, the sensitivity to CPC levels were detected. There was a noted higher level of CPCs in circulation detected in patients with symptomatic MM as compared to the other disease states evaluated. The microfluidic device captures the CPCs and they can be washed off the device and utilized in advanced molecular and cytogenetic techniques for further genetic profiling. Currently, bone marrow biopsy samples are utilized, as well as whole blood, for this testing in patients.
The researchers also pointed out the value of this device in its cost to the healthcare system. Prior chip iterations have had significant cost, rendering this method of analysis as too much of a financial burden to healthcare and patients to be implemented broadly. The particular chip they have reported designed is made of plastic and can be injection molded quickly and inexpensively. This increases its chances of making further on the market and being utilized broadly.
Proponents of this technology share that the bone marrow biopsy will no longer be needed but it is still a gold standard for diagnosis and provides a glimpse at the health of an individual’s bone marrow and immune system. There may definitely be a future for this technology for MM, and other cancers, especially as it relates to sparing a patient the agony of a biopsy; however, other information is gleaned from that biopsy than just the presence of plasma cell expansion.
Sources: Integrative Biology, University of Kansas News,