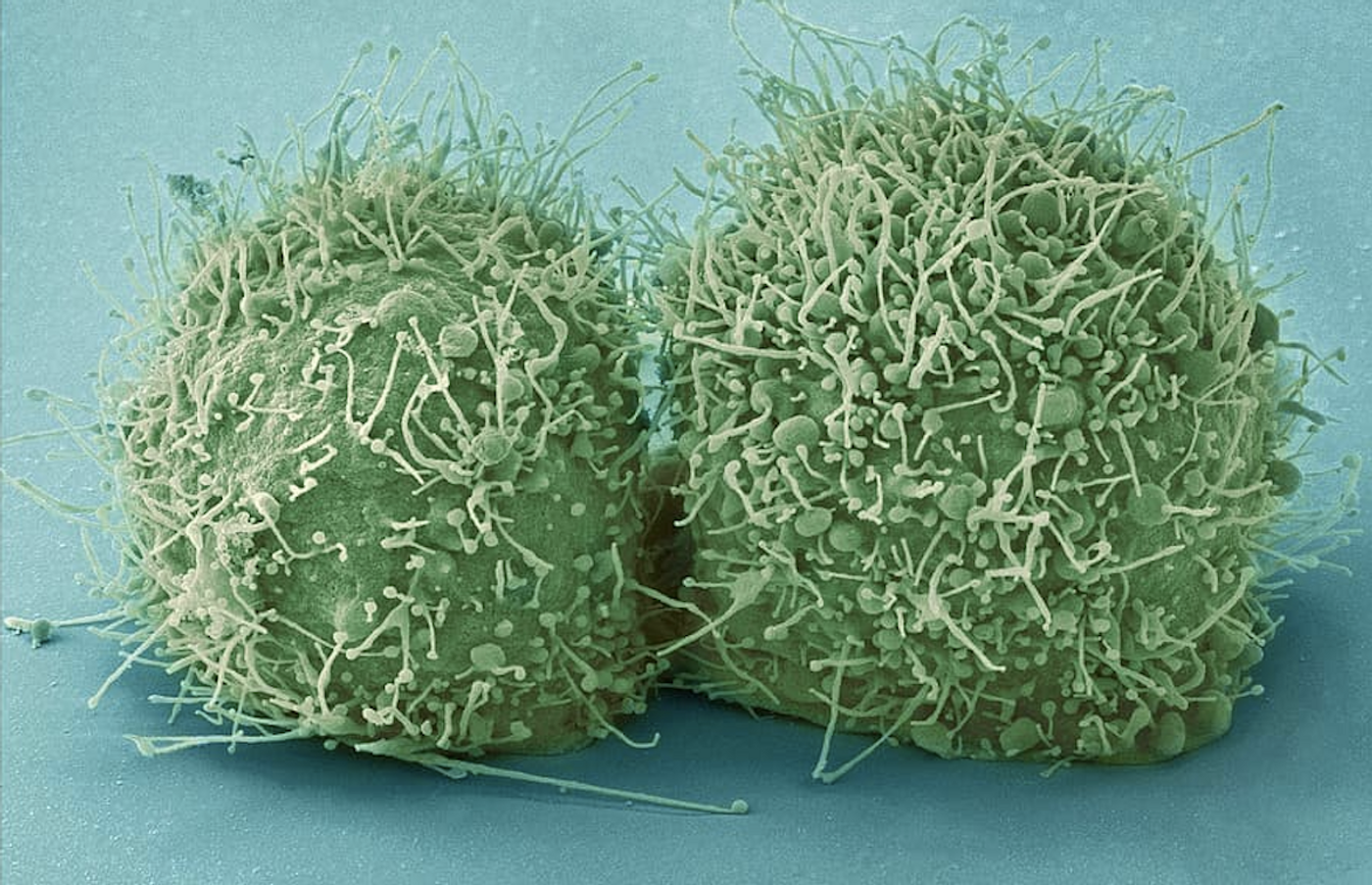
Cilia are Found to Have Vesicles That May Influence Heart Disease
Cells have many specialized organelles, including a kind of signaling hub called a cilium, which sticks out of the surface of most cell types like an antenna. Defects in cilia can cause serious diseases called ciliopathies that impact many different organ systems, including the kidneys and heart. Researchers have now learned more about the cilium and how problems with ciliary proteins can contribute to heart disease.
Chapman University postdoctoral researcher Ashraf Mohieldin, Ph.D., and colleagues used imaging at the level of individual cilia and proteomic studies to investigate a swelling in the ciliary membrane called a ciliary bulb. The ciliary bulb was found to have similarities to other structures in the cell called extracellular vesicles. The findings have been reported in Advanced Science.
Extracellular vesicles are little sacs that can carry cellular signals from one to cell another, facilitating communication. The ones associated with cilia called ciliary extracellular-like vesicles (cELV), were found to contain 172 proteins. These proteins were shown to be involved in ciliary signaling and the maintenance of homeostasis in the heart in zebrafish and mouse models. The cELV also appears to be an ejectable structure that can be released with fluid force.
"Our findings reveal for the first time crucial ciliary proteins that are implicated in ciliopathy disorders," said Mohieldin. "We hope that our research will rejuvenate our understanding and current approach to investigate human genetic diseases."
When cELV proteins were eliminated from the zebrafish, the researchers observed many abnormalities that have been associated with ciliopathies, including cystic kidney disorders, hydrocephalus, and random heart looping. An examination of heart contractility showed that reduced cELV levels in both zebrafish and mice cause a reduction in heart muscle tension, disrupting heat function along with left ventricular hypertrophy and cardiac fibrosis. This caused a high mortality rate in mice.
In mice that lacked cELVs, there was a significant reduction in ejection fraction, stroke volume, and the output of the heart. This is the first study to link cELVs and physiological abnormalities in these models.
Future work will hopefully shed more light on how cELVs influence disease, so that ciliopathy patients can be treated.
"Targeting these proteins can help scientists to clearly understand the mechanism of these disorders and ultimately lead the path to potential treatments for ciliopathies," said Mohieldin.
Sources: AAAS/Eurekalert! via Chapman University, Advanced Science