Heart Organoids with a Contracting Chamber are Created
Scientists that study human disease have long had to rely on animal models or cell culture platforms in which cells grew in a layer in a dish, both of which do not always allow researchers to get an accurate picture of human biology. This problem extends to many research fields, which led investigators to create better cell culture models. Some of these are three-dimensional structures consisting of multiple types of cells that spontaneously organize in ways that mimic simple versions of human organs; these models are called organoids.
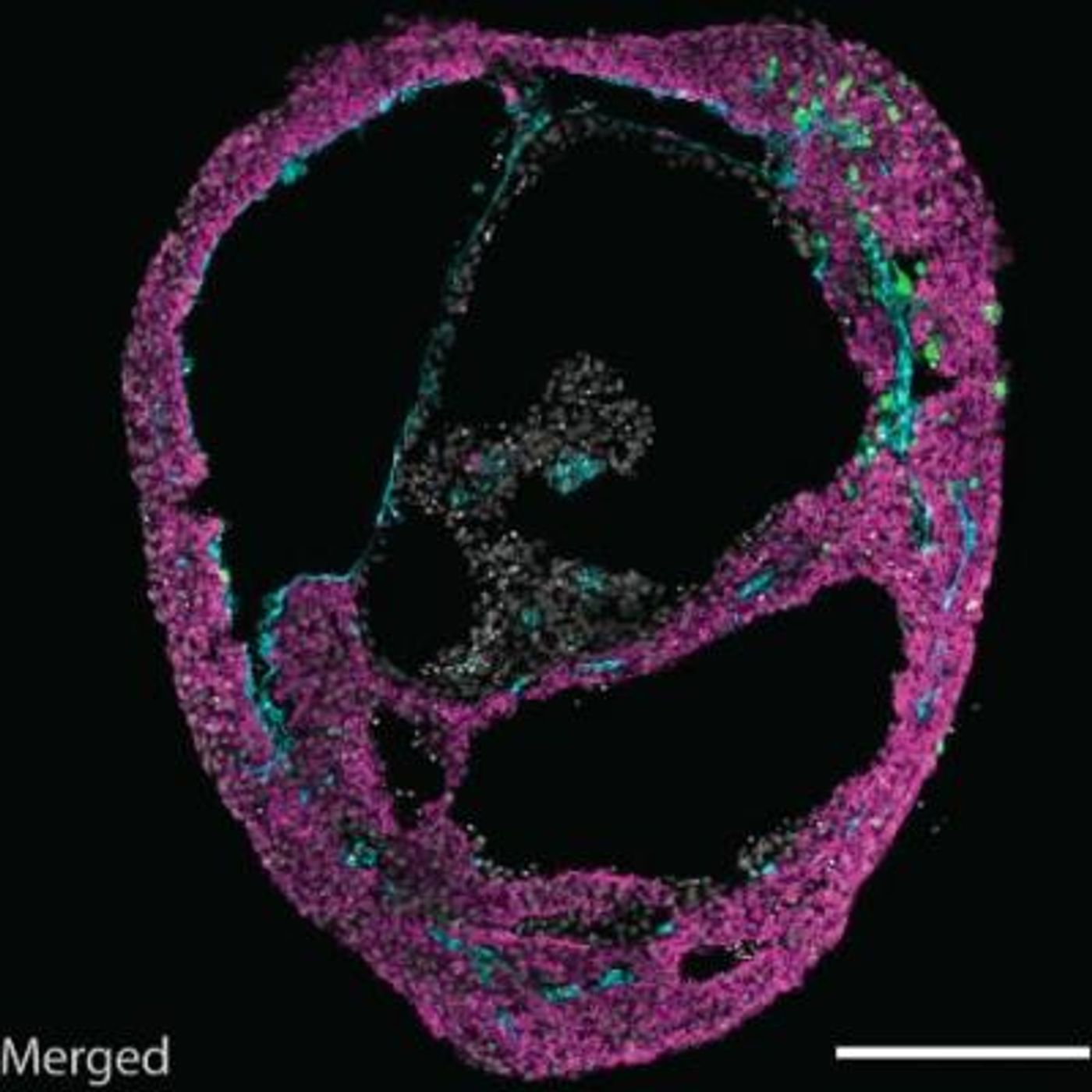
There are human heart organoids, and now, researchers have made them more like real hearts. The scientists guided human pluripotent stem cells to develop into heart models that are about the size of sesame seeds. They can do so without the need for additional scaffolding, which has made these self-organizing cell groups some of the best heart organoids to date. They even have a hollow chamber. The work has been reported in Cell.
Previous approaches have used tissue engineering to attempt to create three-dimensional cardiac organoids. These models, however, have not responded like human hearts do to damage, and thus, they are usually not good for studying disease.
"Tissue engineering is very useful for many things like, for example, if you want to do measurements on contraction," said biologist Sasha Mendjan of the Austrian Academy of Sciences in Vienna. Organs that develop naturally do so with cells that put themselves in the right place and form the proper structures.
"Self-organization is how nature makes snowflake crystals or birds behave in a flock. This is difficult to engineer because there seems to be no plan, but still something very ordered and robust comes out," said Mendjan. "The self-organization of organs is much more dynamic, and a lot is going on that we do not understand. We think that this 'hidden magic' of development, the stuff we do not yet know about, is the reason why currently diseases are not modeled very well."
In this work, the researchers were able to coax heart cells to organize themselves by activating six pathways, which are all known to play a role in embryonic heart development. As the stem cells take on their specialized heart cell identities, they form different layers, including a structure that is like the wall of the heart. About a week into their development, they spontaneously formed a 3D structure with a chamber, similar to human heart development. The wall-like tissue also started to rhythmically contract, which pushed liquid around the inside of the cavity.
"It's not that we are using something different than other researchers, but we are just using all of the signals known," Mendjan explained. All six pathways may not be needed to push stem cells to differentiate into heart cells, he noted. "So they thought, 'Okay, they're not really necessary in vitro.' But it turns out all these pathways are necessary. They are important to make the cells self-organize into an organ."
The researchers also subjected the cardioids to damage to observe their response. When a steel rod was used to simply kill cells, in a mimic of a heart attack in which heart cells simply start dying off, the researchers saw cardiac fibroblasts move to the site of the injury. These fibroblasts began to pump out restorative proteins to repair the damage.
"We want to come up with human heart models that develop more naturally and are therefore predictive of disease," Mendjan said. "This way, companies will be more open to bringing more drugs into the clinical trials because they are much more certain of the outcome of the trial."
The team is planning to make these cardioid models even more complex, and are hoping to add more chambers to the 3D cell model.
Sources: AAAS/Eurekalert! via Institute of Molecular Biotechnology of the Austrian Academy of Sciences, Cell