One of the many methods behind improving treatment for adverse cardiovascular events include helping the heart help itself - through regeneration.
A new study from the University of Houston is studying the potential of equipping the heart with the ability to regenerate cardiomyocytes, the muscle cells of the heart, in response to normal loss due to aging as well as loss from injuries like heart attack. After all, a 2010 study from
BMC Cancer showed that five-year survival rates for heart failure patients is worse than most cases of cancer.
When cardiomyocytes die, they are replaced with connective tissue similar to how a skinned knee will scar over time. While the epidermal tissue will eventually grow back, heart tissue won’t regenerate. Instead, the resulting fibrosis of the heart muscle impairs the ability of the heart to pump, which makes the foundation of why cardiovascular disease is so lethal. If the body tissues don’t have adequate access to oxygen and nutrients via blood, the tissues will die.
The new Houston study, published in the journal
Proceedings of the National Academy of Sciences, researchers found a completely new player in the game of cardiomyocyte formation regulation. Small RNAs, understandably called microRNAs (mRNAs), were believed by the scientists to contain potential for turning scarred tissue back into functional tissue.
"I expect these microRNA regulators to be potent, either acting alone or in combination with the existing agents we have,” said Robert Schwartz, PhD, from the University of Houston. Schwartz and his research team were among the first scientists to make turning fibroblasts into cardiomyocytes a realistic treatment for heart muscle damage.
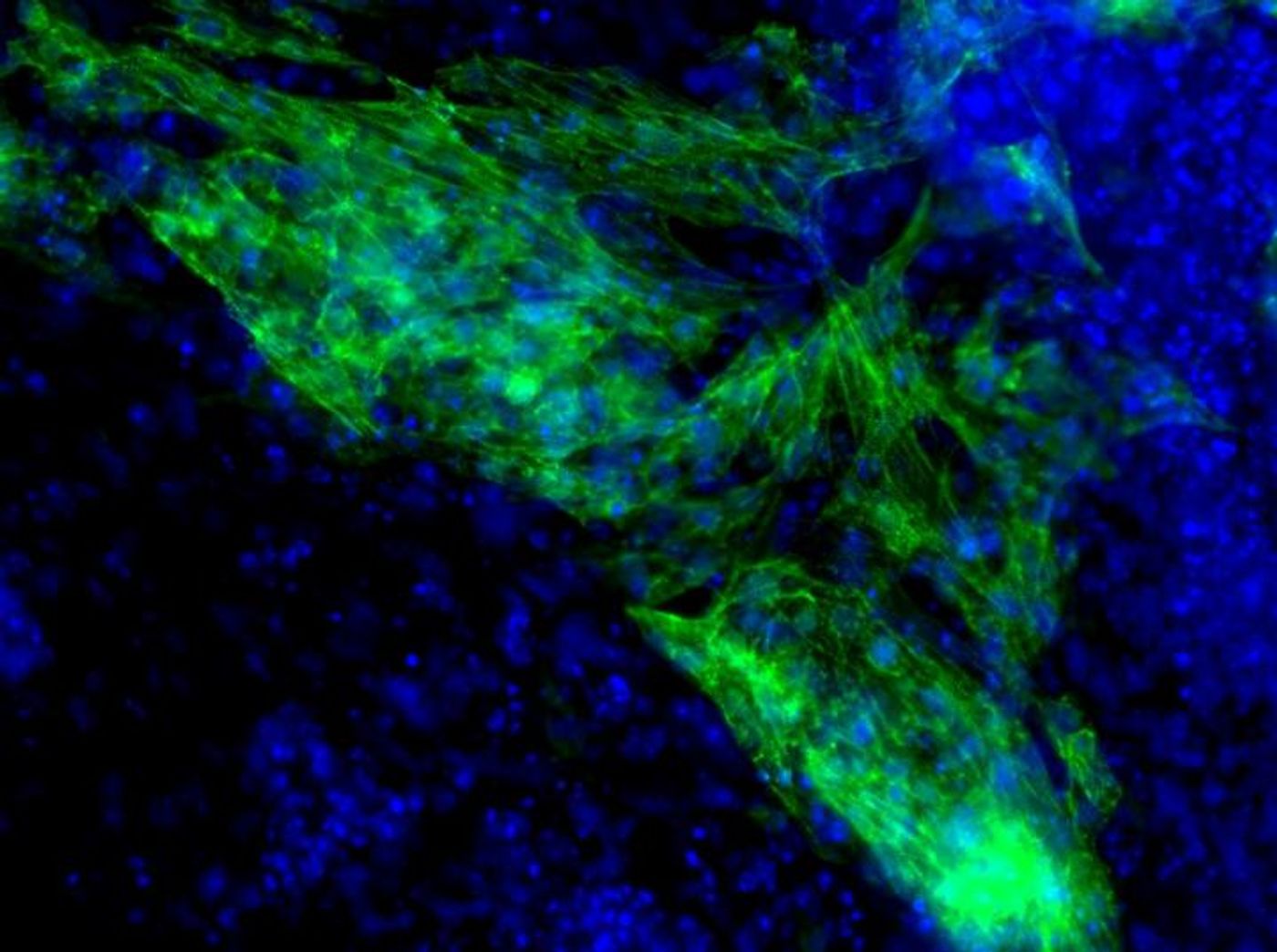
The current Houston study tracked the process of cardiomyocyte formation isolated from mice embryos, looking specifically for a role of mRNAs in gene regulation for cardiomyocyte genes. Can mRNA regulators turn human fibroblasts into heart muscles? They asked themselves. Identifying potential candidates would be a promising step for medical approaches for treating cardiovascular disease. Applying mRNAs clinically would be relatively efficient since mRNAs are active early in the the heart formation process and are easy to deliver into human bodies.
The researchers ultimately identified over 100 mRNAs in the population of early heart cells. They further indicated that a specific mRNA, called the miR-322/503 cluster, appeared to by the best candidate for stimulating cardiomyocyte formation.
In the future, the researchers want to build on their discovery by looking for more mRNAs that could be used to treat heart attacks and heart failure and apply their findings clinically within the next decade. With mouse models for studying the miR-322/503 cluster already in existence, the researchers are hopeful that their findings will be clinically relevant very soon.
Sources:
University of Houston,
BMC Cancer