Injectable Gel Empowers Heart Cells to Regenerate
If human heart cells could regenerate the way salamander or zebrafish cells can, then perhaps cardiovascular disease wouldn’t be the leading cause of death in the United States and worldwide. But for mostly unknown reasons, heart cells - called cardiomyocytes - can’t come back after being damaged. However, a new technique designed by a team of experts from the University of Pennsylvania aims to empower heart cells to become regenerative.
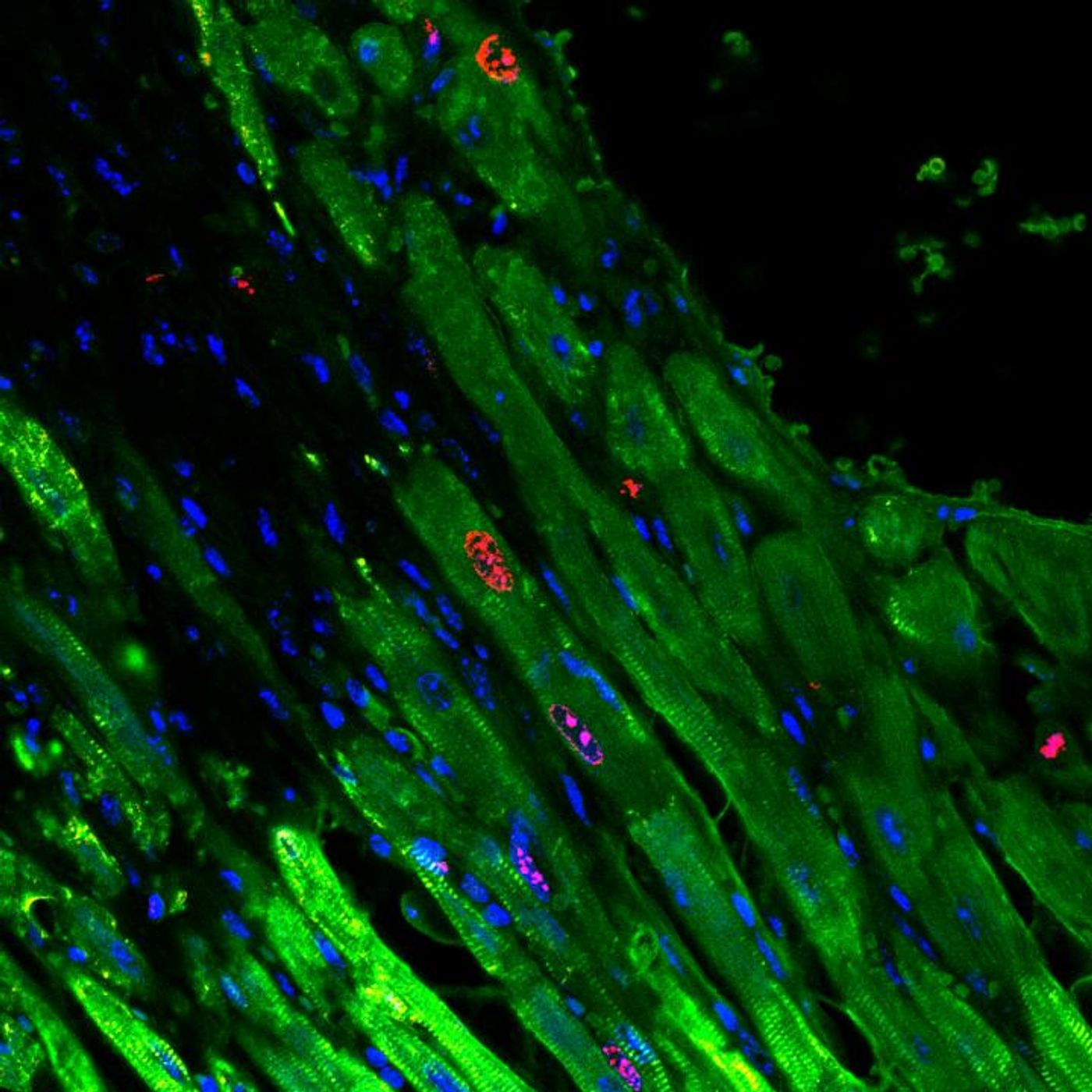
When cardiomyocytes either die or are damaged during a heart attack, the heart is left to do the same job - pumping oxygenated blood to the entire body - but with less cells. The new approach, described in a study published in Nature Biomedical Engineering, is based on an injectable gel that blocks the cellular signals telling heart cells to stop proliferating.
The gel contains and slowly releases microRNAs, short pieces of genetic sequences, directly into the heart muscle. It’s the microRNAs that facilitate interaction with specific signaling pathways that prevent heart cells from proliferating after damage like that from a heart attack. In experiments where mice models of heart attack were treated with the gel, they showed significant improvement during recovery compared to mice who didn’t receive the gel.
The key for applying this technique is in the dosage, explained study leader Edward Morrisey. “The microRNAs that we used last less than eight hours in the bloodstream, so having a high local concentration has strong advantages,” he explained.
This also means that patients would need to be injected frequently and with large doses.
According to study scientist Jason Burdick, the gel contains two key qualities that make it successful in protecting microRNAs and delivering them to the heart muscle:
-
Shear-thinning: “It has bonds that can be broken under mechanical stress, making it more fluid and allowing it to flow through a syringe or catheter.”
-
Self-healing: “When stress is removed, the gel’s bonds reform, allowing it to stay in place within the heart muscle.”
According to graduate student Leo Wang, the gel also features “attachment sites that keep the microRNAs in place. As the gel breaks down, it loses its grip on the microRNAs, which can slip out of the gel and into the cardiomyocytes."
The gel is not without its concerns. The study researchers explain that there is a potential risk of the gel reaching cells other than cardiomyocytes, with the potential of promoting tumor formation. Going forward, researchers will have to thoroughly test the gel and the microRNAs it carries for both its effectiveness and its safety.
"We're seeing a change in approaches for regenerative medicine, using alternatives to stem cell delivery," Burdick said. "Here, instead of introducing new cells that can have their own delivery challenges, we're simply turning on repair mechanisms in cells that survive injury in the heart and other tissues."
Sources: University of Pennsylvania, World Health Organization