Realistic Heart Models Grow Cardiomyocytes
Injecting healthy heart cells grown in the lab is a promising treatment option for people with heart diseases whose own heart cells have died or been damaged. But scientists have had trouble growing heart cells in the lab to completion. Now, using essentially what is a functional model of the human heart, Michigan Technological University scientists are growing fully-functional heart cells.
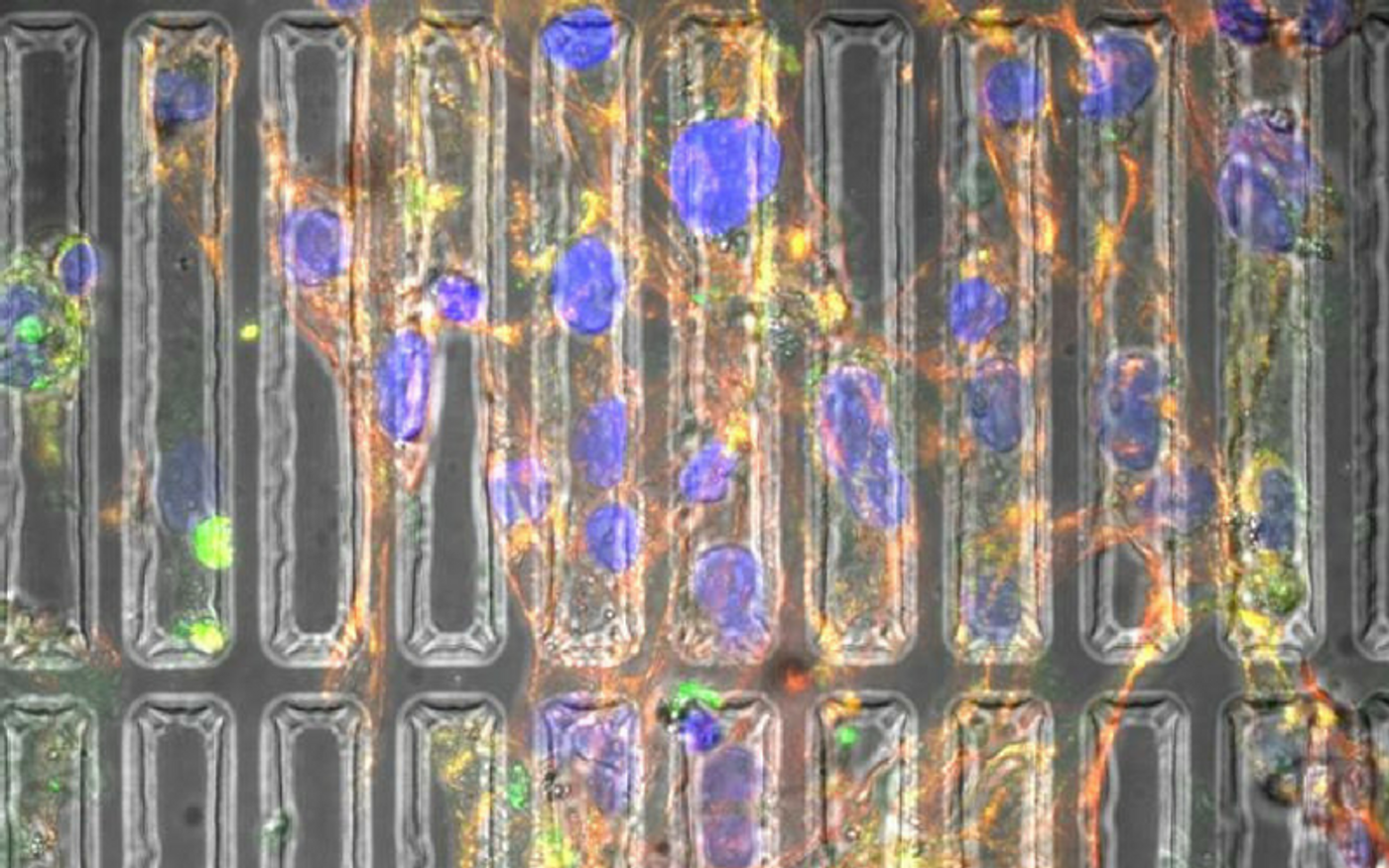
Heart muscle cells, called cardiomyocytes, are grown from pluripotent stem cells in petri dishes in a laboratory. But so far, these lab-grown cardiomyocytes have been underdeveloped, sub-par compared to real human cells. Scientists believe that the problem is the petri dish, simply because a petri dish is not a heart. The petri dish does not capture the pressure and other forces acting on naturally-growing heart cells.
"The mechanical properties of substrates play an important role in the cell behavior because the mechanical cues that cells sense in the actual (heart) environment is unique," explained Parisa Pour Shahid Saeed Abadi. "We are using biochemical and biomechanical cues to enhance the differentiation and maturation.”
Abadi and the team from Michigan Technological University created three-dimensional models of the human heart, designed for the sole purpose of making the growing cardiomyocytes “feel at home” during their maturation. The model was able to recreate biomechanical properties like pressure and stiffness, both natural physical forces that heart cells experience in the body.
By mimicking the environment of the human heart, cardiomyocytes grown in the lab this way were stronger and more mature. "On day one we start seeing the effect of the substrate on the morphology of the cells," Abadi said.
Researchers also found that this growth method led to changes in the cell and in the nucleus, both which alter gene expression, for the better.
Growing cardiomyocytes in a heart-like environment has the potential to improve maturation time and functionality, minimizing the rate of rejection by human bodies. And with this qualities, cardiomyocytes could be realistically used as treatment for heart disease, when an individual’s own cardiomyocytes die or are damaged due to heart attack, stroke, or something else.
Going forward, Abadi and the team are going to continue making enhancements on the substrate preparation methods. Particularly, they are interested in improving communication between cardiomyocytes, another vital feature for their maturation.
The present study was published in the journal Advanced Functional Materials.