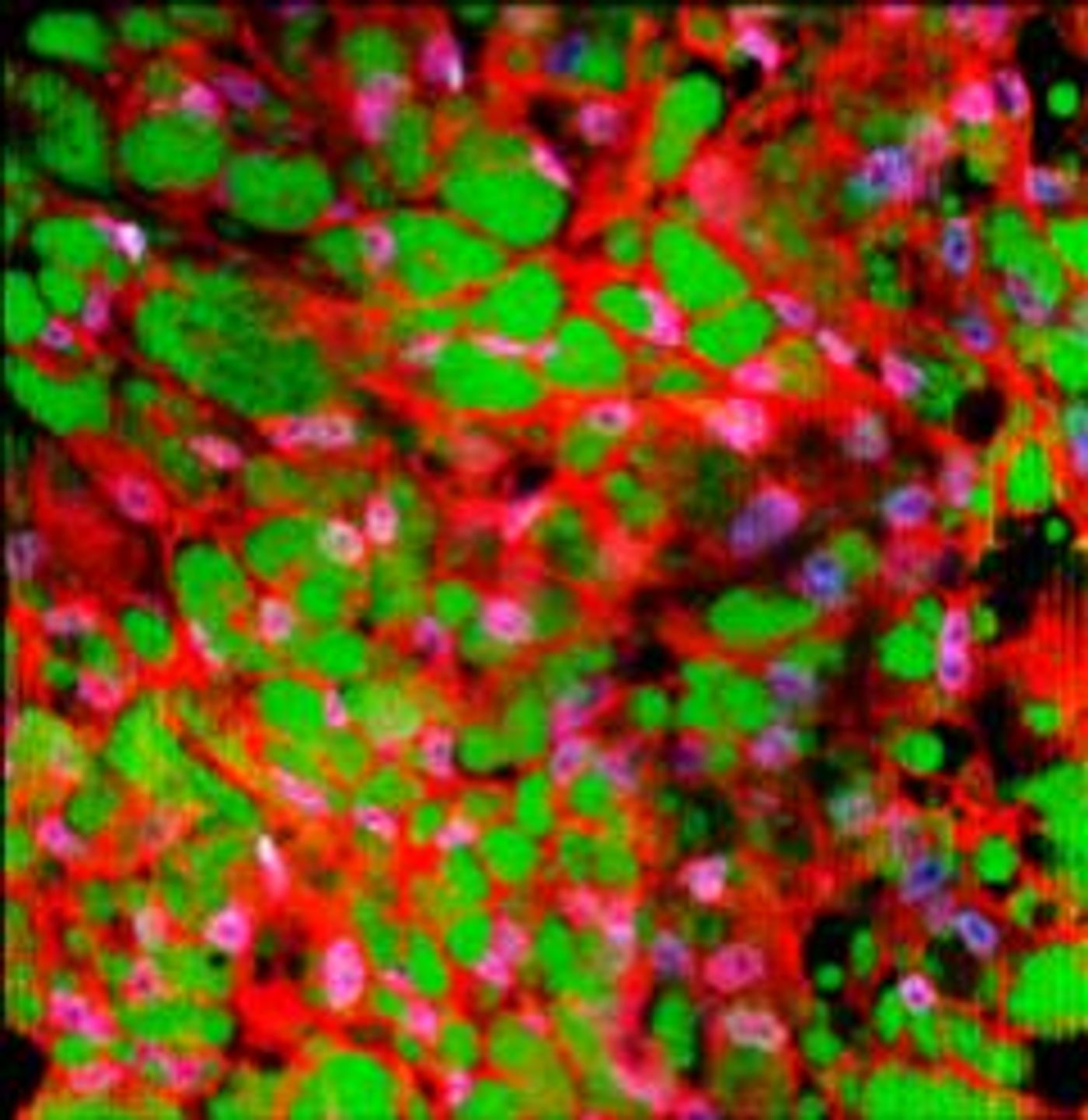
Changing White Fat to Brown Fat
Not all fat acts equally. It’s been established that brown fat can burn energy and generate heat, while white fat is far more common, and can store energy and release hormones. It’s been suggested that brown fat is a healthy fat, and that increasing brown fat levels might help manage weight and reduce diabetes symptoms. That has been a challenge, however.
Researchers at Columbia University have now developed a method to take white fat from a patient, convert it to brown fat in the lab, and re-implant it. This work uses techniques that are commonly utilized by plastic surgeons. The team, led by Sam Sia, has published their work in Scientific Reports; they successfully changed white fat to brown fat and have potentially created a new therapy for patients.
Previous efforts to convert fat have used chronic cold exposure, something most people have a difficult time enduring; a pharmaceutical route has also been created, but drugs can cause side effects.
"Our approach to increasing brown fat is potentially safer than drugs because the only thing going into patients is their own tissue, and it's highly controllable because we can tune the amount of brown fat we inject," said Sia. "The process is also so simple that it could be potentially performed using an automated system within a doctor's office or clinic."
For this work, the researchers used media, or food for cells, that was spiked with molecules to encourage browning, incubating the white fat in that media for one to three weeks to turn it brown. The browning was assessed with biomarkers including UCP1 protein levels. This demonstrated that white fat can be converted to brown fat in the lab. Next, they found that the brown fat remained stable (for two months) when they infected in back into a mouse from which white fat had been taken.
"The persistence of the converted brown fat is very important because we know that when white fat is naturally stimulated to turn to brown fat in vivo, through cold exposure, for example, it can rapidly change back when the stimulation is removed," explained study co-author Brian Gillette, a biomedical engineer now working in the department of surgery at NYU Winthrop Hospital. "Even though we could repeat the procedure several times if we needed to since it's minimally invasive, it is critical that the brown fat survives well and remains stable so that it can function as an effective therapy."
They confirmed that this technique worked on human subcutaneous fat; it was changed to brown fat successfully. "This suggests that it might be possible one day to attempt our approach in humans as a potential therapy to help with weight loss, control of blood glucose levels, or to prevent weight gain," said lead author Nicole Blumenfeld, a graduate student in the Sia lab.
Unfortunately, the mice they tested did not seem to lose weight after their brown fat implant. But this work is a starting point to see if such a technique is useful for promoting dramatic weight loss.
"This is an exciting advance toward engineered brown adipose tissue in clinical applications if it is proven to be safe and effective in humans," added Li Qiang, assistant professor in pathology and cell biology at Columbia University Medical Center who was not a part of this research.
The team is continuing this work to see if it has an impact on metabolism. "There is a clear need to explore new weight-loss approaches with the potential for low rates of complications and long-term efficacy," Sia said. "The ability to culture large quantities of tissue at once while retaining its 3D vascular structure is advantageous and holds promise as a potential approach in clinical weight management."
Lesrn more about the types of fat cells from the video.
Sources: AAAS/Eurekalert! Via Columbia University School of Engineering and Applied Science, Scientific Reports