New Findings on the Early Stages of Autism Spectrum Disorder
While the prevalence of autism spectrum disorder has grown, there is still a lot we don’t know about what causes the developmental disorder. Scientists at Salk Institute have gained new insight into the neurological differences between people with and without ASD. Their study created stem cells from normal and ASD patient samples, which showed that ASD-affected stem cells had significantly different rates and patterns of development compared to stem cells unaffected by ASD. The findings have been reported in Nature Neuroscience, and the researchers are hopeful that this work will help lead to therapeutic interventions that can be used in the early stages of ASD.
"Although our work only examined cells in cultures, it may help us understand how early changes in gene expression could lead to altered brain development in individuals with ASD," said the senior author of the study Rusty Gage, a Salk Professor and President of the Institute. "We hope that this work will open up new ways to study neuropsychiatric and neurodevelopmental disorders."
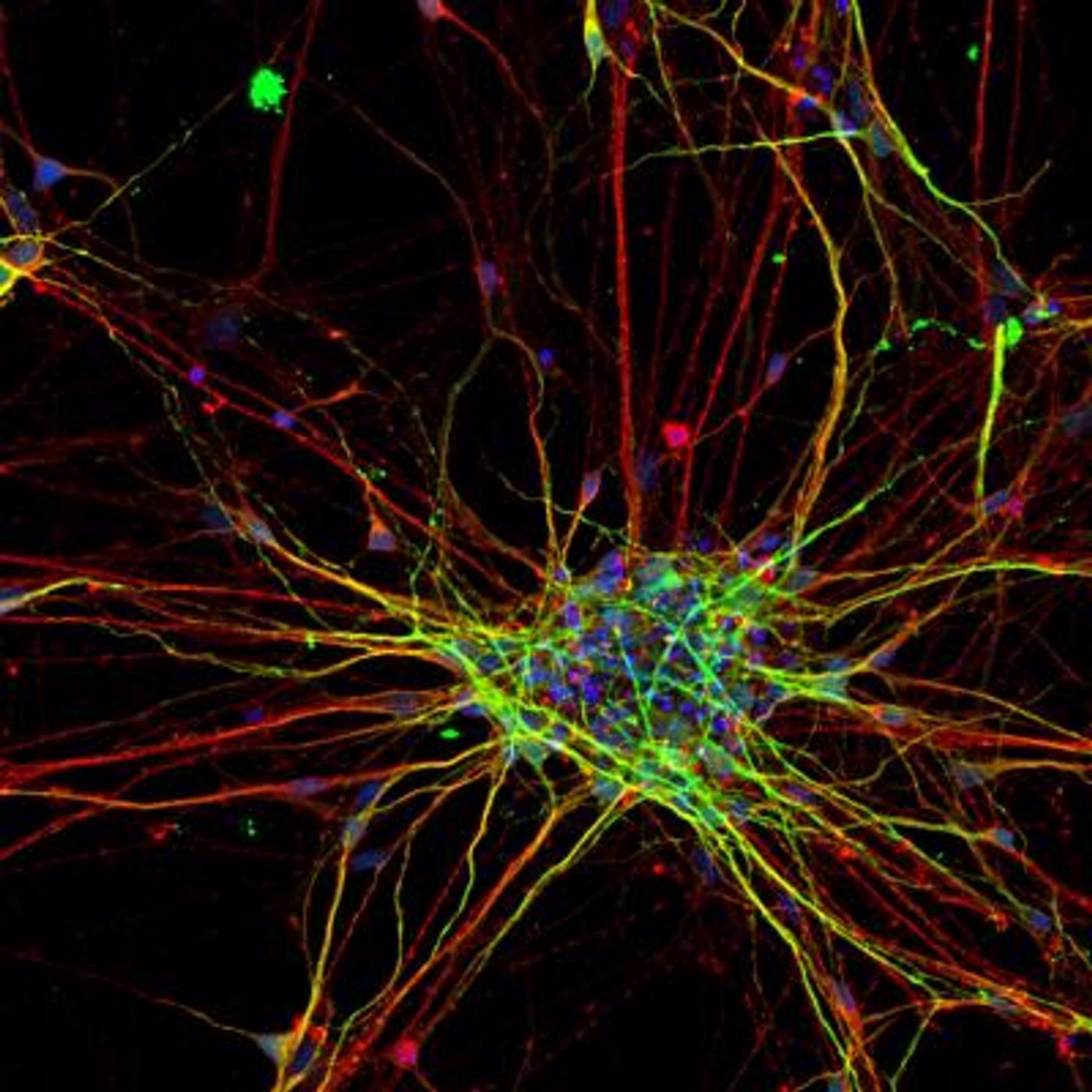
The scientists generated pluripotent stem cells from the skin cells of eight ASD patients and five neurotypical people. Pluripotent stem cells are able to become any cell type, and with various chemical reagents, the researchers coaxed them to become nerve cells.
This effort allowed the team to track the stem cells throughout their developmental stages, identifying the genetic patterns as they become neurons. The stem cells derived from ASD patients were markedly different from the other stem cells; the genetic switches linked to neural stem cell stages were coming on earlier in the affected stem cells. The researchers found that expression changes were occurring in genes that have already been linked to an increase in the risk of ASD. When the nerve cells did develop, they grew more quickly and had a greater degree of complexity in their branching compared to control cells.
"It's currently hypothesized that abnormalities in early brain development lead to autism, but the transition from a normally developing brain to an ASD diagnosis is blurred," said the first author of the work Simon Schafer, a postdoctoral fellow in the Gage lab. "A major challenge in the field has been to determine the critical developmental periods and their associated cellular states. This research could provide a basis for discovering the common pathological traits that emerge during ASD development."
"This is a very exciting finding, and it encourages us to further refine our methodological framework to help advance our understanding of the early cell biological events that precede the onset of symptoms," noted Gage. "Studying system dynamics could maximize our chance of capturing relevant mechanistic disease states."
The investigators think that this work will open up new avenues for the study of mechanisms that underlie the predisposition and development of ASD. They are continuing this work and want to use brain organoids next. Organoids are miniature, simplified versions of human organs, and will enable the researchers to see how different cells interact in models of human ASD-affected brains.
"The current diagnostic methods are mostly subjective and occur after the emergence of behavioral abnormalities in young children," Schafer added. "We hope these studies will serve as a framework for developing novel approaches for diagnosis during an early period of child development--long before behavioral symptoms manifest--to have the maximum impact on treatment and intervention."
The video above demonstrates how brain organoids are grown in the lab.
Sources: AAAS/Eurekalert! via Salk Institute, Nature Neuroscience