Scientists Use CRISPR Gene Editing in Mice Successfully
The gene-editing tool CRISPR/Cas9 was adapted from the bacterial immune system for use in cells growing in culture and in animal models that develop rapidly like zebrafish and insects. Using the tool in mammals like mice presented major challenges, however. For one thing, it takes a long time to produce several generations of mice, which is necessary for studying inherited genetic changes like the ones introduced by CRISPR. Now researchers at UC San Diego have found a way to make it work in a mouse model, and want to use it to edit multiple genes at the same time. Their research, reported in Nature and discussed in the video, creates a foundation on which other investigators can build, which is what’s happened with CRISPR techniques in other research models.
"Our motivation was to develop this as a tool for laboratory researchers to control the inheritance of multiple genes in mice," explained Cooper. "With further development we think it will be possible to make animal models of complex human genetic diseases, like arthritis and cancer, that are not currently possible."
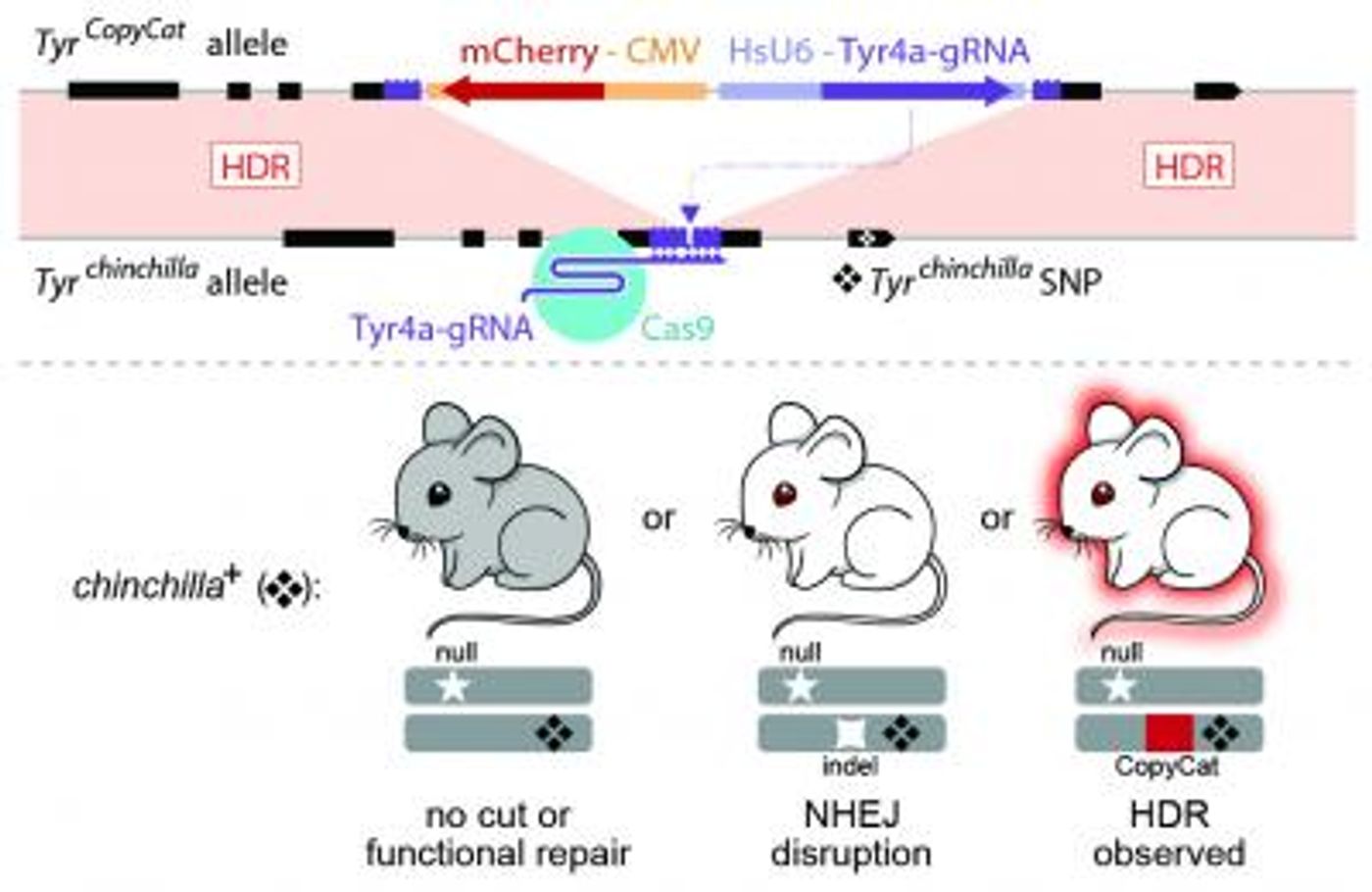
For this work, the scientists inserted a genetic element called CopyCat into a gene that regulates fur color. If CopyCat impacts both copies of the fur color gene, black fur turns white - an obvious way to see if the genetic engineering attempt has been successful.
Using a variety of approaches, the team led by UCSD Assistant Professor Kimberly Cooper showed that when CRISPR/Cas9 cuts the genome, the CopyCat element can move to repair that break. The genetic element could be easily added to an additional chromosome. The team found that 86 percent of offspring inherited the element, instead of 50 percent. This strategy worked if used in female mice during egg production, but not in males producing sperm.
The researchers are already using their technique to study the insertion of other traits and genes. "We've shown that we can convert one genotype from heterozygous to homozygous. Now we want to see if we can efficiently control the inheritance of three genes in an animal. If this can be implemented for multiple genes at once, it could revolutionize mouse genetics," said Cooper.
This work has the potential to be used to investigate a variety of questions, including complex human traits and diseases, among other things. "We are also interested in understanding the mechanisms of evolution," said Cooper. "For certain traits that have evolved over tens of millions of years, the number of genetic changes is greater than we can currently assemble in mice to understand what caused bat fingers to grow into a wing, for example. So we want to make lots of these active genetic tools to understand the origins of mammalian diversity."
Sources: AAAS/Eurekalert! Via UCSD, Nature