Using Laser Tweezers to Learn More About Protein Droplets
Researchers have been investigating how the movement of liquids inside of cells can affect the processes that are occurring within. Now, scientists have developed a way to manipulate spherical droplets of proteins that form inside of cells and revealed more about how they become more like a gel. It seems that in some conditions, like when a lot of other proteins are around, some of those droplets harden. These properties may have an impact on human health and disease, and now scientists are finding innovative new ways to study them. Their findings have been reported in Biomolecules.
"These droplet-forming proteins are a relatively new area of study, so we know very little about their basic properties," noted lead investigator Priya R. Banerjee, Ph.D., an assistant professor of physics in the University of Buffalo College of Arts and Sciences. "As physicists, we want to quantify the dynamics of these droplets and learn what factors influence them. This is important as the dynamics of protein droplets are a key to their cellular function and dysfunction. Prior research has focused on the structure of the proteins themselves, but our work shows that environmental factors are equally important. We see that external conditions can alter the internal state of the droplets, which may affect their function in human cells."
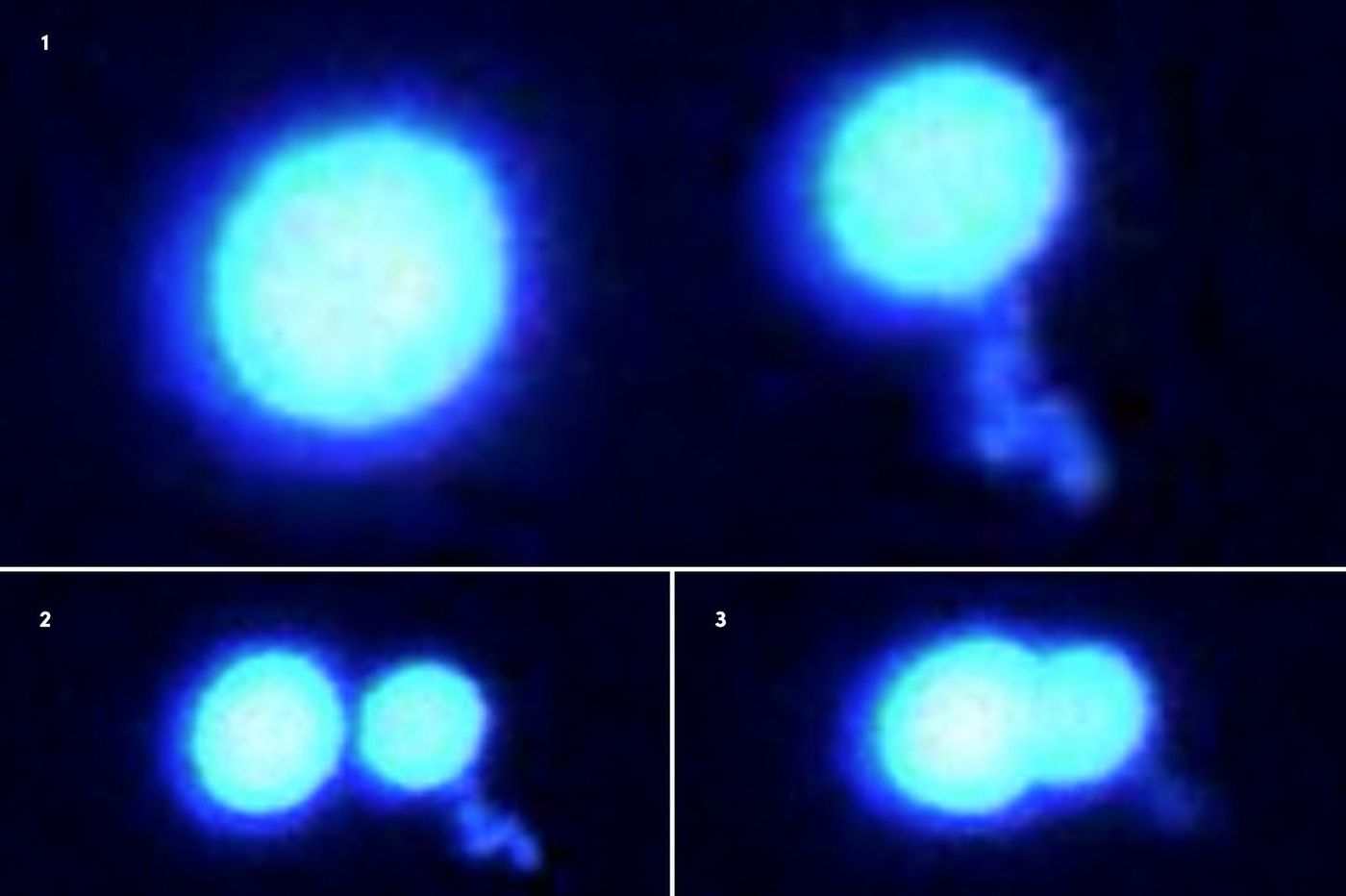
Some research has suggested that dense droplets of proteins influence various cellular functions including stress responses, the immune system, and gene expression. In this research, the scientists focused on a protein, fused in sarcoma (FUS), known to form droplets. While FUS droplets are in liquid form in healthy brains, they clump into solid material in a neurodegenerative condition, amyotrophic lateral sclerosis (ALS). The researchers used optical tweezers, which are highly focused laser beams, to push protein droplets together in a solution.
The droplets of proteins were found to easily merge, generating one larger drop, if other inert molecules, like polyethylene glycol (PEG) crowded the solution. If the levels of PEG or other chemicals in solution went up, the droplets wouldn’t combine completely, and they got more gelatinous. The investigators also used lasers in another way - to study how protein droplets behave in crowded environments.
Banerjee’s team added fluorescent tags to various protein molecules in one droplet. They used a high-intensity laser to poke the middle of the droplet, which caused fluorescent molecules that the laser hit to go dark, permanently.
They also timed new proteins that moved into the darkened area. Protein droplets did so rapidly in solutions that were sparsely populated, but the process was far slower when the solution was crowded with chemicals. That indicated to the researchers that protein droplets get more viscous, and tougher to move through when their environment is crowded.
"Our experiments were done in test tubes, but our results suggest that inside living cells, the crowding status could affect the dynamics of protein droplets," Banerjee says.
The scientists have yet to determine if droplet fluidity is related to the clumping seen in ALS patients. Banerjee wants to know more about it.
Sources: AAAS/Eurekalert! Via University of Buffalo, Biomolecules