Learning More About the Significant Roles of Tiny Proteins
The genes in our genome encode for proteins, and different cell types express different genes, which give them their specific functions and capabilities. The letters of protein-coding genes are translated into strings of amino acids that are folded into a three-dimensional structure. Some proteins can be hundreds or even thousands of amino acids long, but there are some that are much shorter. Scientists are beginning to learn more about very small proteins that are composed of a string that’s less than 100 amino acids long; these small proteins are sometimes called microproteins.
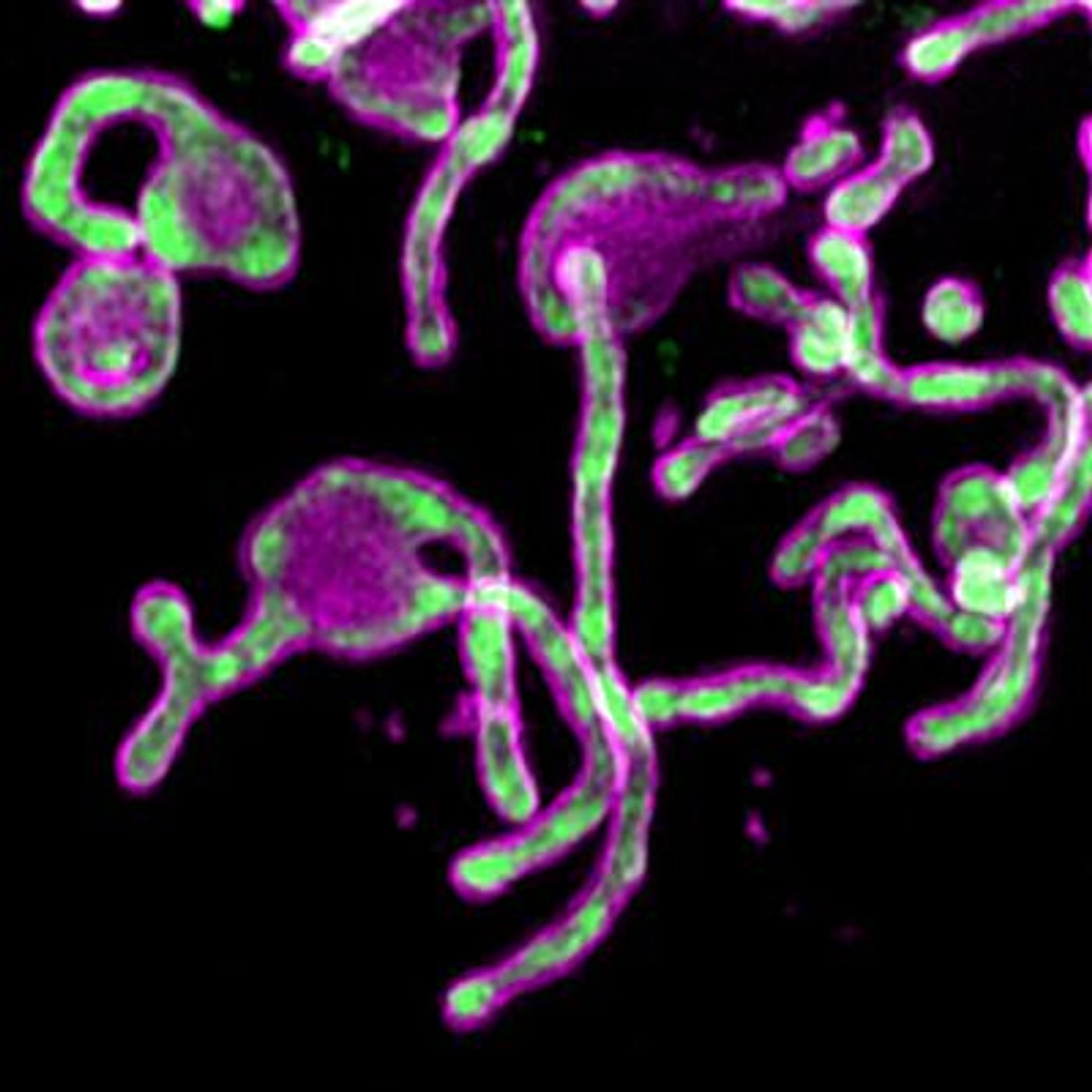
Scientists at the Salk Institute have been investigating one microprotein in particular, called PIGBOS, which is made up of only 54 amino acids. Research has recently shown that PIGBOS is linked to relieving cellular stress. The protein was identified in mitochondria, organelles that generate energy for the cell, by Salk postdoctoral researcher Qian Chu, though it had been difficult to find the protein or reveal its role. While many researchers add fluorescent tags to proteins to learn more about their function and location in cells, that approach is challenging to use with such small proteins; for example, the tag can be too large for the small protein or change how it works.
When Uri Manor, director of the Waitt Advanced Biophotonics Core Facility at Salk, tried to add a common fluorescent tag called GFP to PIGBOS, there were problems. So instead of adding the entire GFP tag, the team split the GFP into smaller components and only added a portion of it, called the beta strand.
The scientists were then finally able to learn more about what PIGBOS was doing. Reporting in Nature Communications, the investigators found that PIGBOS resides on the mitochondria’s outer membrane. There, it interacts with another protein called CLCC1, which is part of the membrane of a different organelle called the endoplasmic reticulum (ER).
"PIGBOS is like a connection to link mitochondria and ER together," said Chu. "We hadn't seen that before in microproteins and it's rare in just normal proteins."
Their work indicated that PIGBOS and CLCC1 work together to control ER stress. When PIGBOS is lost, there is more stress in the ER, which causes more problems for the cell. The ER is a critical organelle that helps fold proteins into their proper shape, and as stress increases, there are greater numbers of misfolded proteins that the cell has to remove; if it doesn’t the cell may die. ER stress and misfolded proteins have been associated with disease. This suggested to the researchers that PIGBOS may be a good therapeutic target.
"This study is exciting because cell stress is important in a number of different diseases, including cancer and neurodegeneration," said the co-corresponding author of the report, Salk Professor Alan Saghatelian. "By understanding the mechanisms behind these diseases, we think we'll have a better shot at treating them."
"Going forward, we might consider how PIGBOS is involved in [a] disease like cancer," noted Chu. "In cancer patients, the ER is more stressed than in a normal person, so ER stress regulation could be a good target."
The team also suspects that other mitochondrial proteins may be involved in ER stress. They also want to learn more about other microproteins.
"Microproteins represent a fledgling field," said Saghatelian. "But I think this work has really impacted our understanding [of] the impact that microproteins can have on biochemistry and cell biology."
"PIGBOS represents one of a limited set of microproteins that anyone has gone through the effort to characterize. And lo and behold it actually has a very important role," added Manor.
Sources: AAAS/Eurekalert! via Salk Institute, Nature Communications