Telomerase Enzyme Found to Have a Protective Role
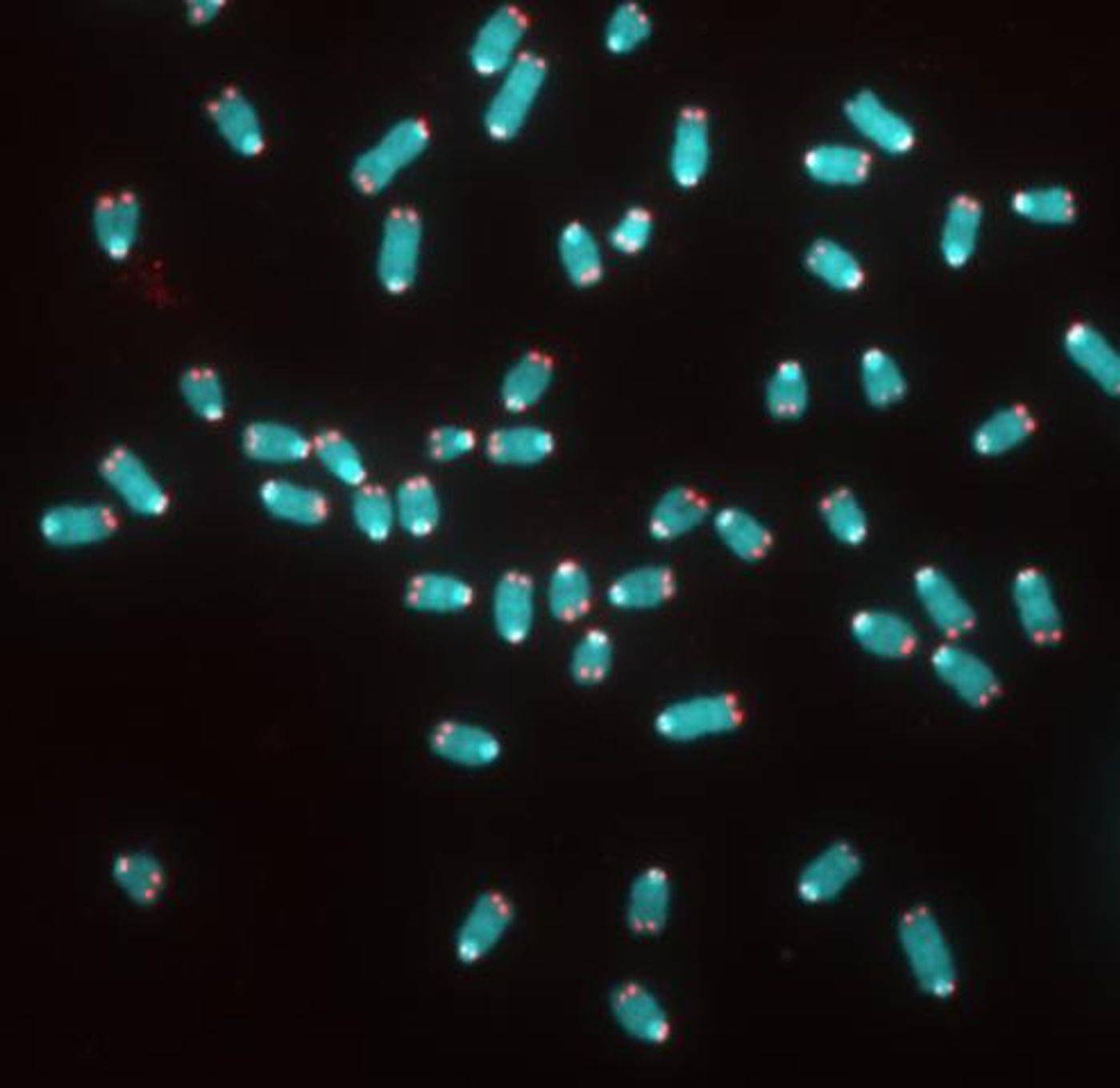
Our DNA is organized into chromosomes, and the ends of chromosomes are protected with caps called telomeres; when cells divide, they lose a very small amount of DNA from those ends. Short telomeres have been linked to aging and disease. An enzyme called telomerase helps maintain those caps by ensuring that the telomeres don't get too small, and helps regulate the cell cycle. The enzyme was thought to be active primarily in cells that divide a lot, like adult stem cells, sperm cells, and cells of the immune system. The enzyme is also activated in tumor cells. New research has found, however, that telomerase has another role.
Reporting in the Proceedings of the National Academy of Sciences (PNAS), the researchers found that in normal adult cells, telomerase is reactivated during one point in the aging process. Right before a cell dies, telomerase appears to help reduce the stress of aging, which slows the process down and reduces damage in DNA that might lead to cancer.
"This study reshapes the current understanding of telomerase's function in normal cells," said the senior author of the report, Kan Cao, an associate professor of cell biology and molecular genetics at the University of Maryland. "Our work shows, for the first time, that there is a role for telomerase in adult cells beyond promoting tumor formation. We can now say that regulated activation of telomerase at a critical point in a cell's life cycle serves an important function."
The telomerase enzyme is crucial during embryonic development, while stem cells are specializing into the many types of cells that are needed to form an organism and cells are rapidly dividing. In healthy adult cells, telomerase is inactive; telomeres get shorter with every round of cell division until they reach a certain length, and cells die. If they don't die, they may take on errors in the DNA, which are a first step toward cancer. It had been thought that telomerase activity in adult cells was only associated with the unlimited cell division that is seen in tumors.
In the Cao laboratory, the scientists worked with mice that were progeny of normal mice and mice that lacked telomerase. The sibling mice all had abnormally short telomeres, but mice that did not inherit the gene that produces telomerase from their normal parents had spontaneous cancer and shortened lifespans compared to the mice that had inherited it.
The researchers found that while skin cells from both mice had short telomeres that were about the same length, the cells from mice lacking telomerase stopped dividing sooner and had a greater likelihood of developing cancer. In mice that still had some telomerase, as the telomeres reached a certain length, a burst of telomerase became active and lessened cancer-promoting DNA damage. Cao suggested that this helps soften the blow of aging by making cell death a more gradual process. In the cells without telomerase, the researchers could prolong their division and reduce damage to DNA by reactivating the telomerase gene.
The findings were confirmed in human cells, with cells lacking telomerase getting shorter telomeres faster, and acquiring more DNA damage compared to cells with telomerase. Now, the researchers want to learn more about how telomerase activity is triggered when telomeres get short.
Sources: AAAS/Eurekalert! via University of Maryland, PNAS