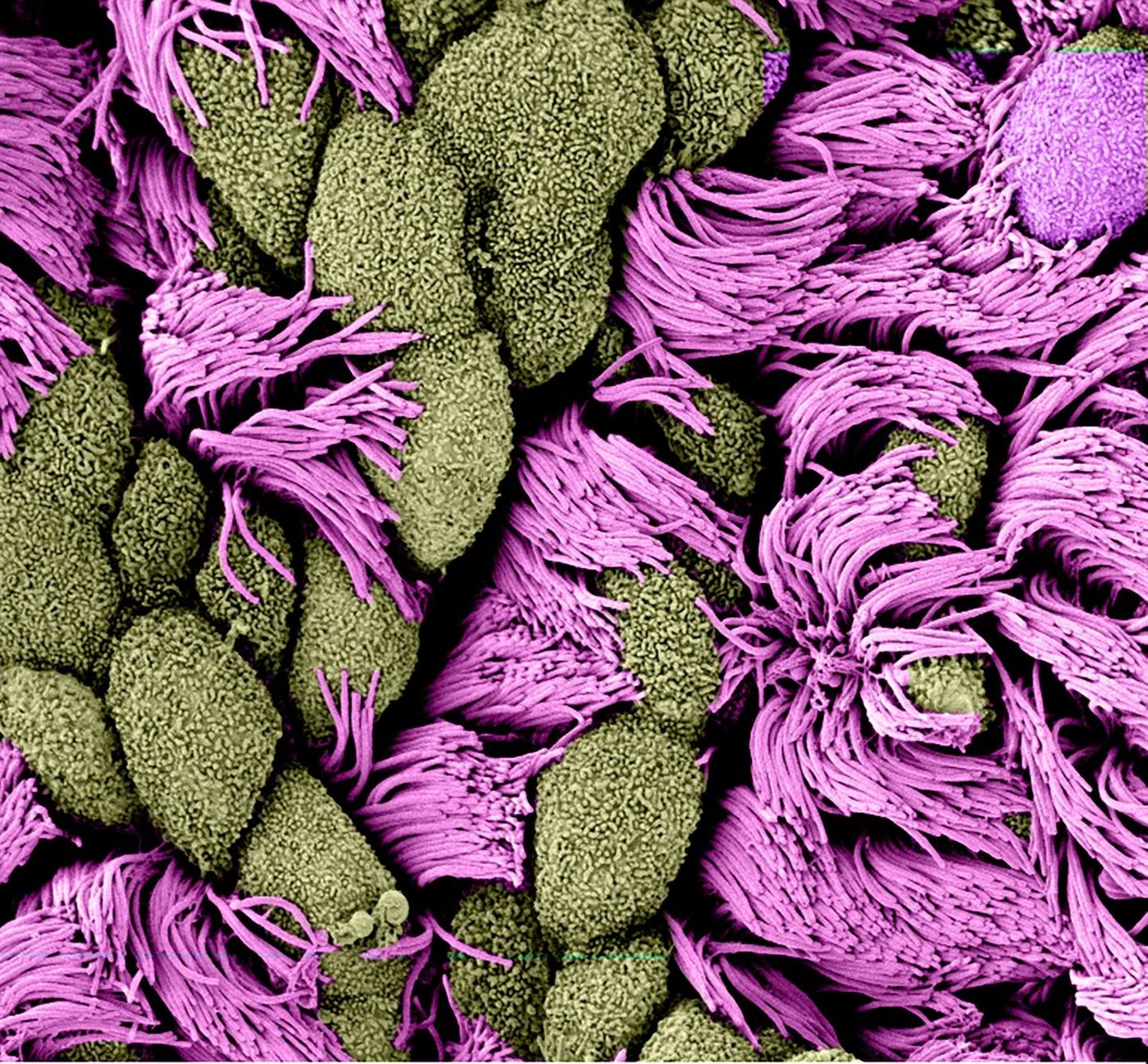
Learning More About How Cells Control the Production of Cilia
Most cell types have an appendage that appears to act as a kind of signaling hub; called a cilium, it's capable of detecting and sending chemical messages. Some specialized cells don’t only carry one of these cilia, however; in the lining of the reproductive and respiratory tracts, cells have hundreds of cilia that move in unison. This ciliary beating helps move fluids through these tracts. Now researchers have learned more about how the cell controls the number of cilia it carries. The findings have been reported in Nature Cell Biology.
"Our main question was how these multicilliated cells become so dramatically different than the rest of the cells in our body," said Andrew Holland, Ph.D., an associate professor of molecular biology and genetics at the Johns Hopkins University School of Medicine. "Most cells make exactly one cilium per cell, but these highly specialized cells give up on this tight numerical control and make hundreds of cilia."
Holland’s team took a close look at a cylindrical structure that sits at the base of the cilium and anchors it, called a centriole. In cells with only one cilium, Holland noted, these centrioles are generated before cell division. There are two parent centrioles in a cell, which are both duplicated so that after cell division, the new daughter cells each get a pair. The older of the two centrioles goes on to form the cilium’s base.
In cells with multiple cilia, special structures called deuterosomes are formed, which become a kind of copy machine that churns out hundreds of centrioles. Thus, these cells can then form many cilia.
"Deuterosomes are only present in multicilliated cells, and scientists have long thought they are central for determining how many centrioles and cilia are formed," added Holland.
The researchers engineered a mouse model that did not carry the gene that encodes for deuterosomes, and then observed what happened in cells that are typically multiciliated. They were surprised to learn that the same number of cilia were present in the multiciliated cell types in the mice that lacked the deuterosome gene. Cells that lacked deuterosomes could still generate new centrioles as fast as cells that had them. They concluded that deuterosomes were not responsible for the huge numbers of cilia.
Next, the team created a mouse that lacked both deuterosomes and parent centrioles, and took a look at the normally multiciliated cells. "We figured that with no parent centrioles and no deuterosomes, the multicilliated cells would be unable to create the proper number of new cilia," explained Holland. However, even the deletion of parent centrioles did not impact cilia number.
"This finding changes the dogma of what we believed to be the driving force behind centriole assembly," noted Holland. "Instead of needing a platform to grow on, centrioles can be created spontaneously."
While it’s rare in mammals, some species, like a type of flatworm, can generate centrioles spontaneously. Additional work in mice showed that centrioles can be created in a part of the cell where there are lots of proteins that make up the centriole; the area is rich in fibrogranular material.
Holland suggested that this area of the cell, about which little is known, carries the crucial parts of centrioles, and can control the number of cilia that are made. Other things, like deuterosomes, are not strictly needed.
"We think that the deuterosomes function to relieve pressure on the parent centrioles from the demands of making many new centrioles, freeing up parent centrioles to fulfill other functions," said Holland.
Knowing more about cilia will help researchers and clinicians treat disorders related to the cilium, so-called ciliopathies. Learn more about them from the video.
Sources: AAAS/Eurekalert! via Johns Hopkins Medicine, Nature Cell Biology