New Microscopy Tools Reveal More About the Role of Actin
Cells rely on a network of tiny filaments to give them form and support their structure. One crucial filament is a protein called actin. Researchers have now devised a new way to monitor the movements of a type of actin as cells move. This cutting-edge imaging technique enabled the scientists to observe actin as it helps a critical organelle in the cell, the powerhouse called the mitochondrion, to divide. The findings have been reported in Nature Methods (the preprint is available here at bioRxiv).
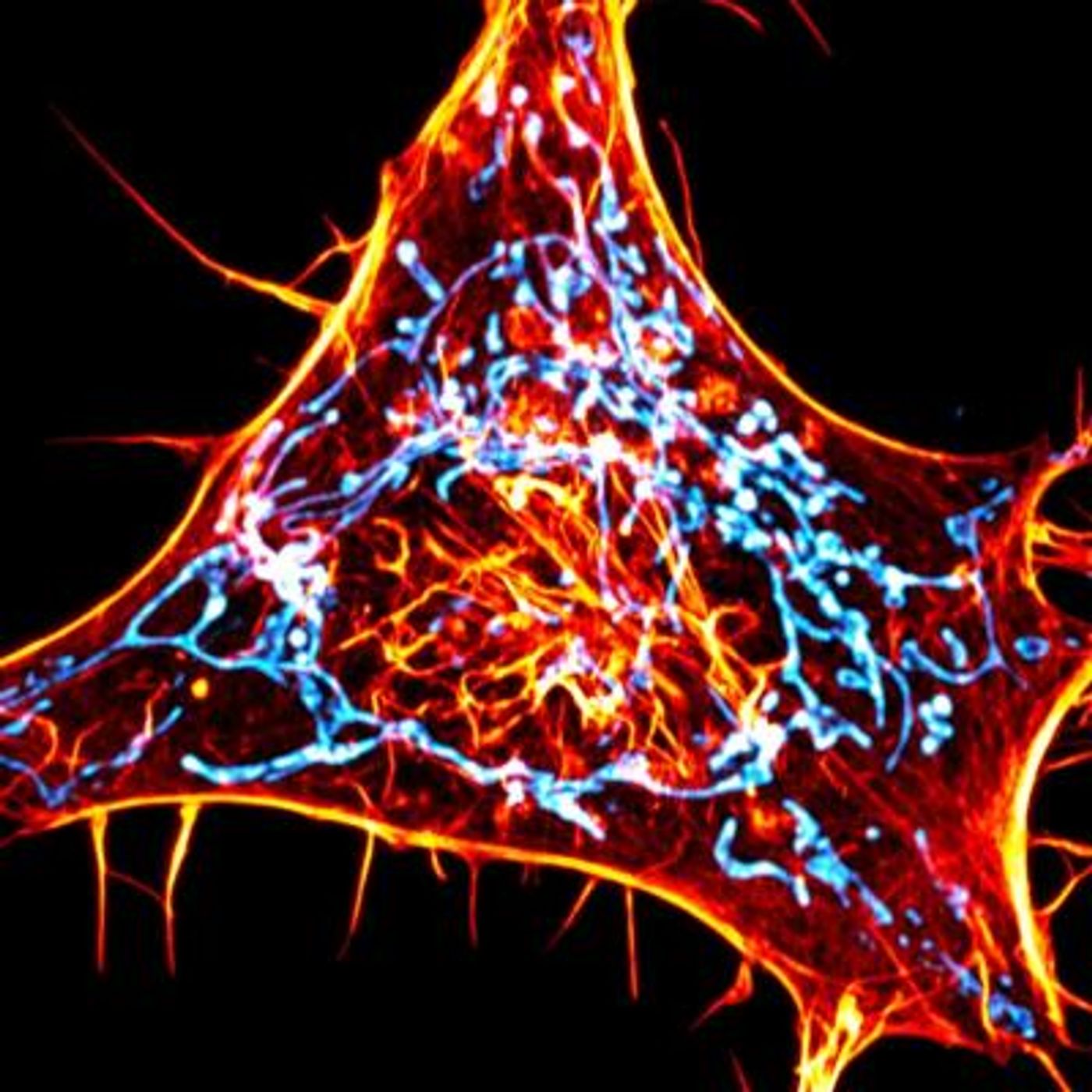
"Actin is the most abundant protein in the cell, so when you image it, it's all over the cell," said corresponding study author Uri Manor, director of the Salk Institute's Biophotonics Core facility. "Until now, it's been really hard to tell where individual actin molecules of interest are, because it's difficult to separate the relevant signal from all the background."
Mitochondrial division, also called fission, occurs during normal maintenance in the cell; they don't only divide along with a dividing cell, they also undergo fission when cells are stressed or mitochondria have sustained damage. While research has suggested that actin is involved in mitochondrial fission, it's been difficult to elucidate its exact role.
In this work, researchers set out to observe actin, rather than trying to remove it from a cell to study its importance - actin is important so that when that happens, the cell is usually ruined. Instead, an actin probe was engineered so that when it's sitting within ten nanometers of mitochondria, a sensor causes it to emit a stronger fluorescent signal.
This study showed that there were bright spots of actin interacting with the mitochondrial membranes. This was also happening where another cellular organelle, the endoplasmic reticulum, was also in contact with the mitochondria, potentially where they were undergoing fission.
Actin hotspots were lighting up and disappearing over time. The scientists showed that there was actin fluorescing within 97 percent of mitochondrial fission sites. They suggested that it was probably happening at 100 percent of fission sites, but the other three percent just weren't visible.
"This is the clearest evidence I've ever seen that actin is accumulating at fission sites," said study co-first author Cara Schiavon, a joint postdoctoral fellow in the labs of Uri Manor and Salk Professor Gerald Shadel. "It's much easier to see than when you use any other actin marker."
Further study suggested that actin links up with the mitochondria before it arrives at the endoplasmic reticulum. They may be coordinating fission together. Other experiments indicated that other organelles like endosomes and lysosomes also divide near endoplasmic reticulum-associated actin.
"This is a universal tool that can now be used for many different applications," said co-first study author Tong Zhang, a light microscopy specialist. "By switching out the targeting sequence or the nanobody, you can address other fundamental questions in cell biology."
Manor is featured in the podcast available above.
"We're in a golden age of microscopy, where new instruments with ever-higher resolution are always being invented; but in spite of that there are still major limitations to what you can see," added Manor. "I think combining these powerful microscopes with new methods that select for exactly what you want to see is the next generation of imaging."
Sources: AAAS/Eurekalert! via Salk Institute, Nature Methods