Why Are Egg Cells So Large?
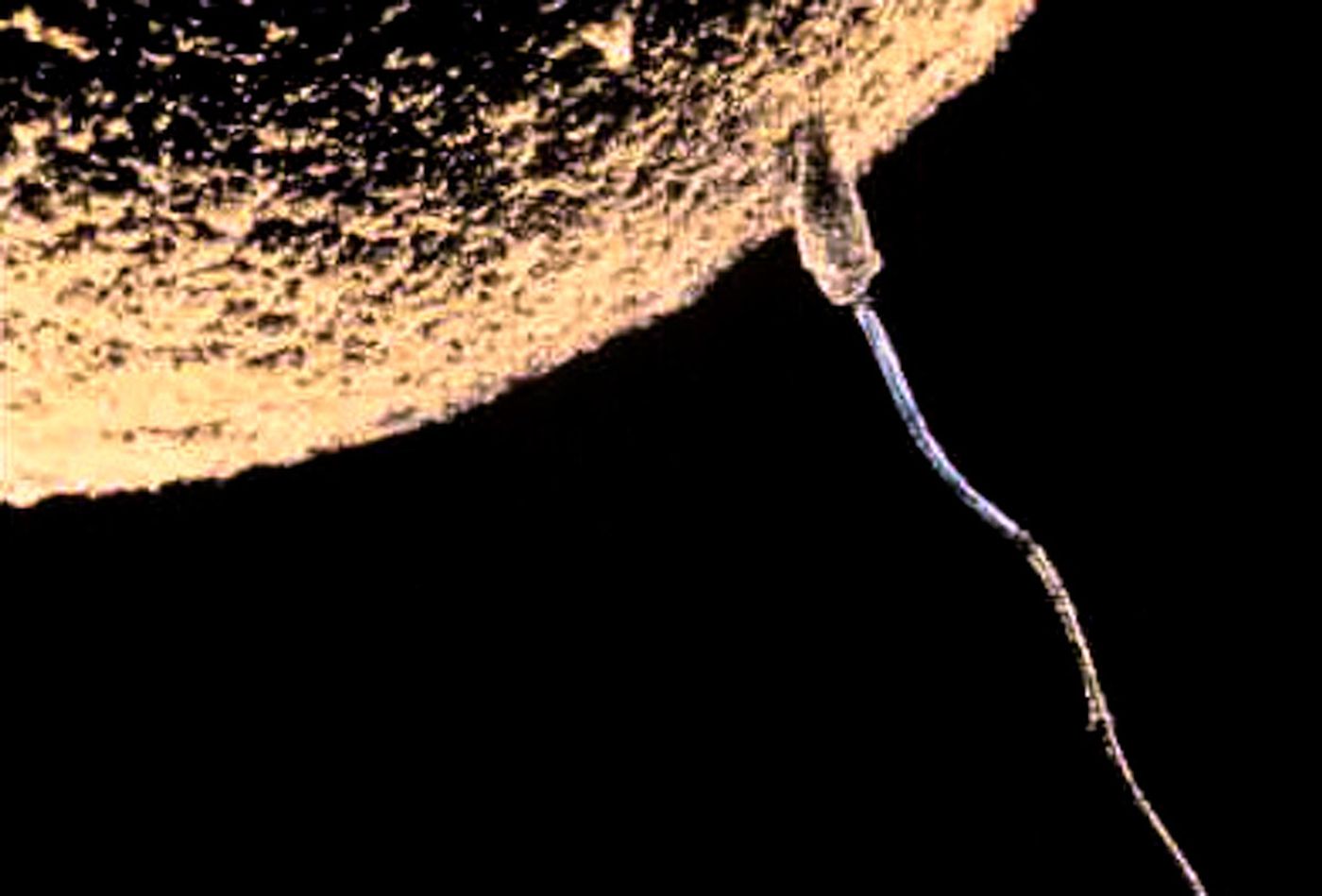
If you've ever seen a video of a sperm cell fertilizing an egg cell, you've probably noticed the huge size difference. That's partly because sperm cells are so small; they only contain 23 chromosomes in a nucleus, some mitochondria, and a few other parts. But egg cells, or oocytes, are also massive; they are several times the size of an average cell in the body and are around 10,000 times the size of sperm. Egg cells are among the biggest body cells in part because after they're fertilized, they have to support the production of an embryo. But there's more to it than that.
New research reported in the Proceedings of the National Academy of Sciences using a fruit fly model has suggested that an oocyte can grow quickly and significantly before fertilization. This process seems to depend on 'nurse' cells that surround the larger oocyte and pour their contents into it, like the exchange of gases between different-sized balloons.
"The study shows how physics and biology come together, and how nature can use physical processes to create this robust mechanism," said co-senior study author Jörn Dunkel, an MIT associate professor of physical applied mathematics. "If you want to develop as an embryo, one of the goals is to make things very reproducible, and physics provides a very robust way of achieving certain transport processes."
In the lab of co-senior study author and MIT associate professor of biology Adam Martin, researchers study myosin proteins, which can function like little motors and are involved in muscle contraction. They started investigating egg development in fruit flies, which involves the formation of egg clusters called cysts. They found that immature oocytes divide into an egg cell and 15 nurse cells, without separating the cells completely. Instead, the cells are connected by little channels that enable the movement of stuff from one cell to another.
They used high-resolution imaging to reveal two phases of transport, and myosin was only playing a role in the second part.
"The two phases are strikingly obvious," Martin says. "After we saw this, we were mystified, because there's really not a change in myosin associated with the onset of this process, which is what we were expecting to see."
Dunkel's lab studies the physics of flowing matter, and they wondered if the cells acted like balloons of different sizes when connected; air flows from a smaller balloon to a larger balloon. The bigger curves on the smaller balloon have more surface tension, so the curvature acts to force the air out and into the larger balloon. "It's counterintuitive, but it's a very robust process," Dunkel said.
The researchers adapted mathematical equations that describe this effect to the cell-based scenario. Nurse cells closest to the oocyte release their contents into the oocyte first, but in two phases, followed by the next closest nurse cell until the most distant is reached.
In the first phase, the channels between the cells get bigger and cytoplasm can move through. Once they're down to about a quarter of their initial size, the second phase kicks in, when myosin contractions push the rest of the nurse cell's contents into the oocyte.
"In the first part of the process, there's very little squeezing going on, and the cells just shrink uniformly. Then this second process kicks in toward the end where you start to get more active squeezing, or peristalsis-like deformations of the cell, that complete the dumping process," Martin said.
The researchers are working to learn more about how the second phase is triggered.
It's too soon to say whether egg cells might be growing this way in other animals. "There's evidence in mice that the oocyte develops as a cyst with other interconnected cells, and that there is some transport between them, but we don't know if the mechanisms that we're seeing here operate in mammals," Martin said.
Sources: AAAS/Eurekalert! via Massachusetts Institute of Technology, Proceedings of the National Academy of Sciences