A Model of Early Human Development is Created
Researchers that study in vitro fertilization, human development and the diseases that may arise during that time, and miscarriages face a lot of challenges in investigating the biology underlying these issues. It's considered unethical to experiment on the human embryos that might be available for such studies, and there are also legal issues. Scientists have now made a breakthrough on this front; they have shown that it's possible to use reprogrammed cells to self-organize into structures that resemble human embryos.
During very early development, a fertilized egg undergoes many rounds of cell division that ultimately generate a structure called the blastocyst, which is composed of several different cell layers surrounding a central mass of cells. The blastocyst is what implants in the uterine wall and goes on to form the embryo.
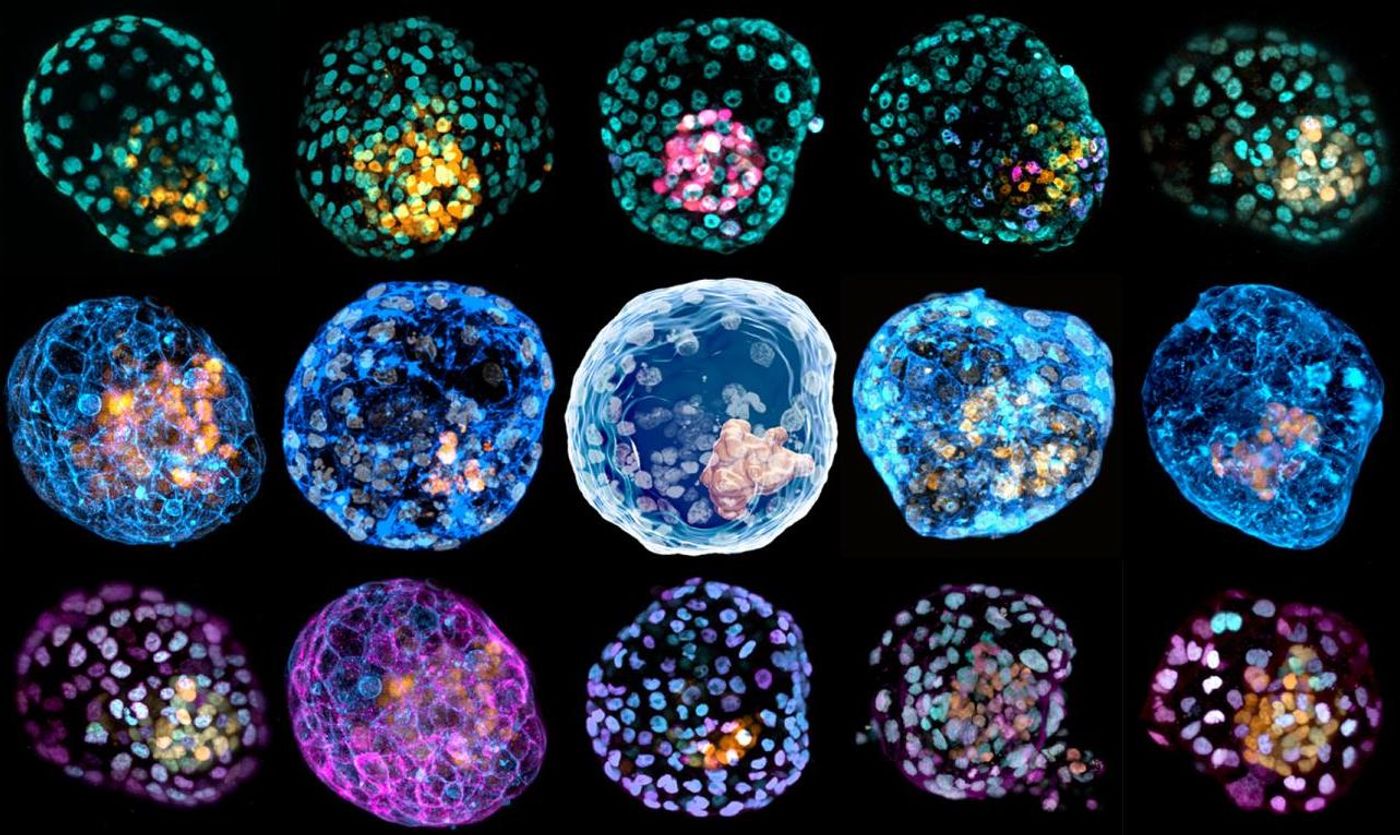
Reporting in Nature, teams used reprogrammed skin cells called fibroblasts to generate three-dimensional human blastocyst models they called iBlastoids.
"iBlastoids will allow scientists to study the very early steps in human development and some of the causes of infertility, congenital diseases and the impact of toxins and viruses on early embryos without the use of human blastocysts and, importantly, at an unprecedented scale, accelerating our understanding and the development of new therapies," noted the study leader Professor Jose Polo of Monash University in Melbourne.
The researchers used "nuclear reprogramming," in which the identity of the cells is changed by altering gene expression. These cells are then put into stuff that can act as scaffolding for the growing cells, like an extracellular matrix. The reprogrammed cells self-organize on this matrix into iBlastoids, which mimic the form and genetics of human blastocysts, including their laters and central mass.
While blastocysts naturally give rise to an embryo, "iBlastoids are not completely identical to a blastocyst. For example, early blastocysts are enclosed within the zone pellucida, a membrane derived from the egg that interacts with sperm during the fertilization process and later disappears. As iBlastoids are derived from adult fibroblasts, they do not possess a zona pellucida" noted Polo.
The researchers did not grow these cellular structures beyond eleven days, which aligns with an international guideline stating that human blastocysts cannot be cultured beyond day fourteen. That guidance may soon be updated to reflect new advances in technologies that don't actually use embryos, but are cell models.
"We are really amazed that skin cells can be reprogrammed into these 3D cellular structures resembling the blastocyst," added co-first study author and Polo lab graduate student Jia Ping Tan.
The researchers are hopeful that this study will provide insights into failed implantation or a lack of progress in development during the first two weeks of pregnancy. The iBlastoids can also help scientists learn more about the early stages of pregnancy.
Sources: AAAS/Eurekalert! via Monash University, Nature