Insight Into an Antiviral Enzyme That Can Impact Cancer Cells
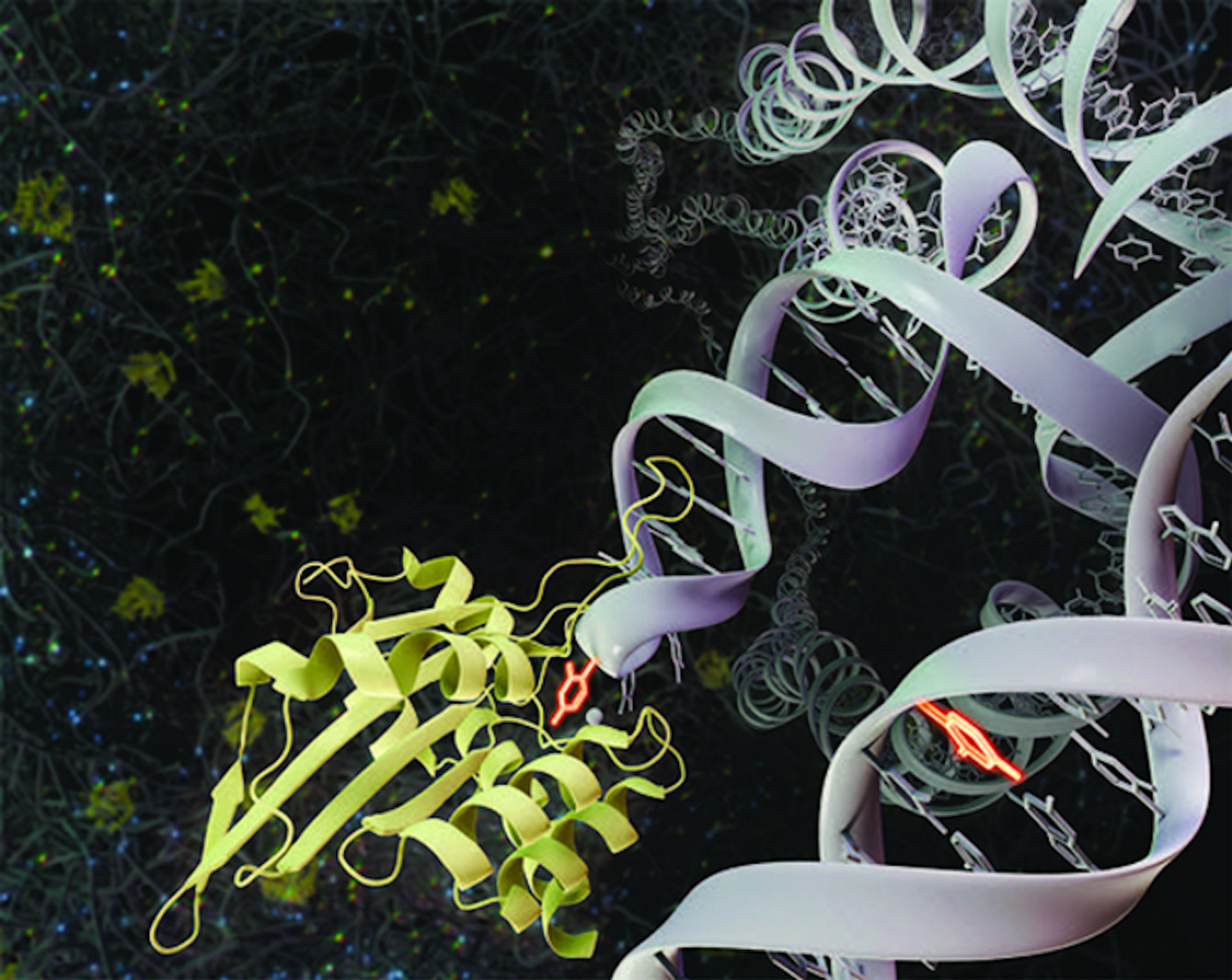
For many years, researchers have been investigating how a family of enzymes called APOBEC3 can help protect the body from pathogenic viruses, and how it relates to cancer. APOBEC3s are known to prevent some genetic changes, like the insertion of retroelements. Scientists have also found that one enzyme in this family called APOBEC3A can trigger the formation of mutations in the genomes of cancer cells. However, this attack may also cause mutations that increase the growth, metastasis, or drug resistance of cancer cells.
“In our previous studies, we demonstrated that APOBEC3A-induced DNA mutations are very frequent in cancer patients. In fact, we found they are present in up to 80 percent of certain cancer types such as lung, breast or bladder cancers,” said Rémi Buisson, Ph.D., an assistant professor at the University of California Irvine School of Medicine.
Researchers have now learned more about how APOBEC3A is expressed in tumor cells. Reporting in Nature Communications, the study authors exposed these cells to chemotherapeutic drugs, which can cause genomic damage and an increase in APOBEC3A levels. When cells were infected with viruses, another increase in the level of APOBEC3A was observed. However, this upregulation of APOBEC3A occurred through two totally different mechanisms.
Viral infection stimulates the innate immune system to respond by activating APOBEC3A, which can eliminate the infection. APOBEC enzymes can coax amino groups off of cytosine that change the moleucle to uracil on either single-stranded DNA or RNA, which generates mutations in viral genomes. That can prevent the replication of the virus.
But the chemotherapeutic drugs trigger APOBEC3A through a different pathway, which may also initiate genomic mutations in the cell that makes the cancer more aggressive. Unlike the virally-induced APOBEC3 response, interferons and a molecule called STAT2 are not involved. Instead, the study indicated that the p65/IkBα pathway plays an important role.
“Together, our results reveal different ways for the cells to regulate APOBEC3A expression to address different types of stresses that the cell may encounter,” said Buisson. “By understanding how cancer cells and viral infections regulate APOBEC3A expression, we are poised to take critical step forward toward the development of both new therapeutic strategies to fight cancer and new antiviral therapies.”
This study suggested that additional work will be needed to learn how to prevent the APOBEC3A-caused mutations in DNA so that tumor growth is not increased.
The researchers also noted that it's possible that APOBEC3A activity may contribute to the formation of mutations in viruses like SARS-CoV-2; more research will be needed to learn how APOBEC3A could be influencing the activity, growth, or mutation of viruses.
Sources: AAAS/Eurekalert! via University of California - Irvine, Nature Communications