Microtubules Found to Act as Mechanosensors
As organisms grow, live, and move through the world, they're subjected to an array of internal and external forces. Biological forms must be able to withstand the stresses of stretching and compression down to the cellular level. Mechanical forces are thought to have a significant impact on the growth, development, and health of cells and tissues.
Cells need a sturdy but flexible framework called a cytoskeleton to give them structural integrity. To perform all the functions that might be required of it, a cell also has to have the ability to move things around the cell. Cellular components called microtubules help perform both functions; they make up part of the cytoskeleton, and they also play a critical role in intracellular transport by acting as a kind of rail network. Motor proteins called kinesin and dynein shuttle things on this microtubule network using molecular fuel called ATP to power their movement. Microtubules are dynamic too; they can grow, shrink, bend, and flex with compression, stretching, and movement.
Now researchers have shown that microtubules can also function as a kind of mechanosensor that can pick up on the state of the cell and adjust transport in the cell accordingly.
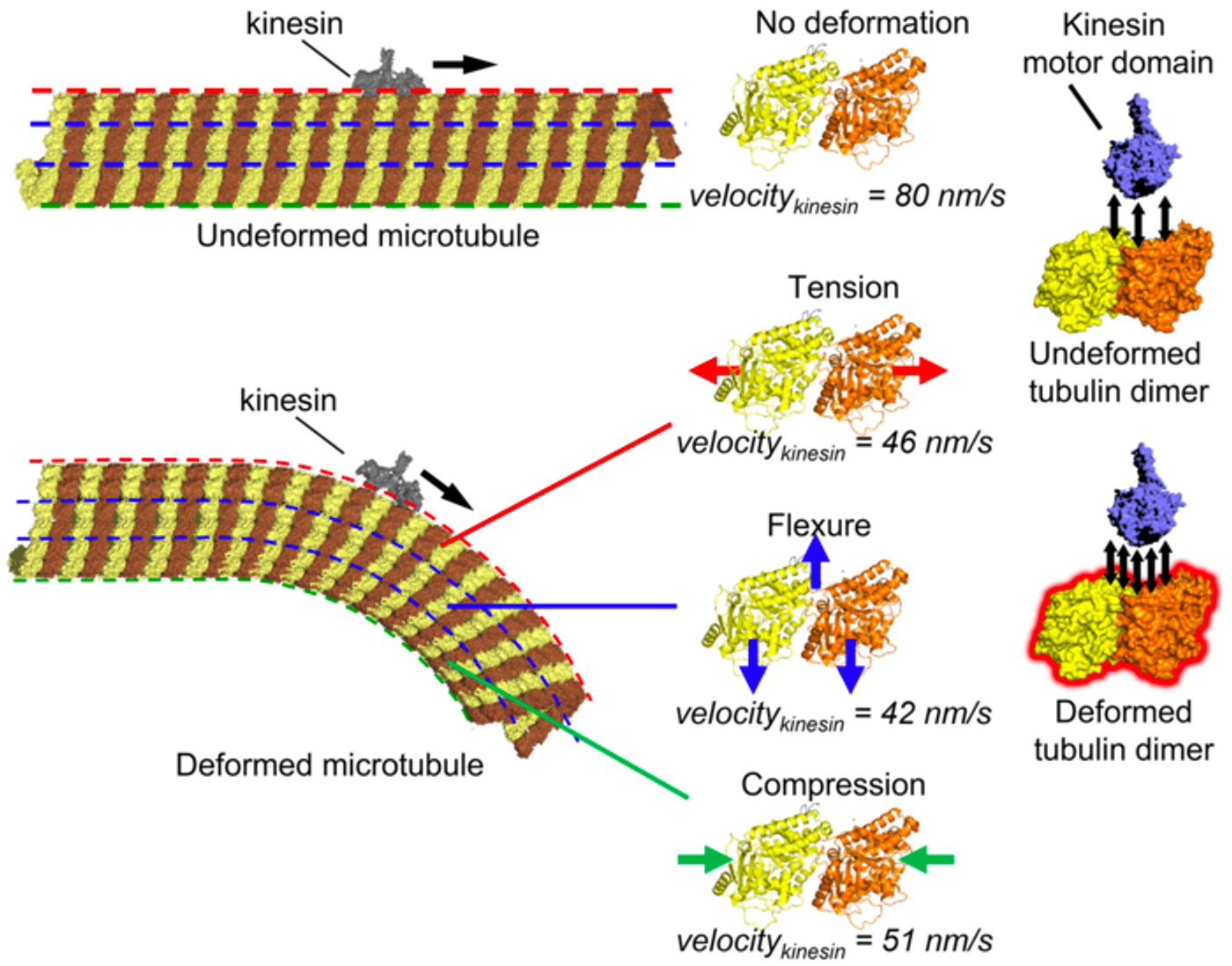
In this study, the researchers used atomic force microscopy and a cell-free model in which they observed individual kinesin motor proteins as they migrated along microtubules that were maintained on a lipid bilayer. The microtubules were kept straight or bent to study how the speed of the kinesin proteins changed under the different conditions, as shown in this the video. When microtubules are bent, the kinesin proteins don't move as quickly. The variability in the speed is illustrated in the image. It had been thought that kinesin gets slower because of cracks or holes in the microtubule structure, but the researchers found that the slowdown occurs even there are no cracks.
The research, which has been reported in Science Advances, suggested that the kinesin proteins seem to stick to the bent microtubules more than they do to straight microtubules. A molecular simulation indicated that the deformed units of microtubules have a greater interaction energy with kinesin, revealing how the conformation of microtubules can regulate the movement of these motor proteins.
This work, which was completed by an international team of researchers, could have implications for biomechanics, biosensors, research that involves the mechanical forces that occur inside and outside of cells, diseases related to intracellular transport or the cytoskeleton.
Sources: Hokkaido University, Science Advances