How A Dangerous Microbe Can Survive for Months Without Water
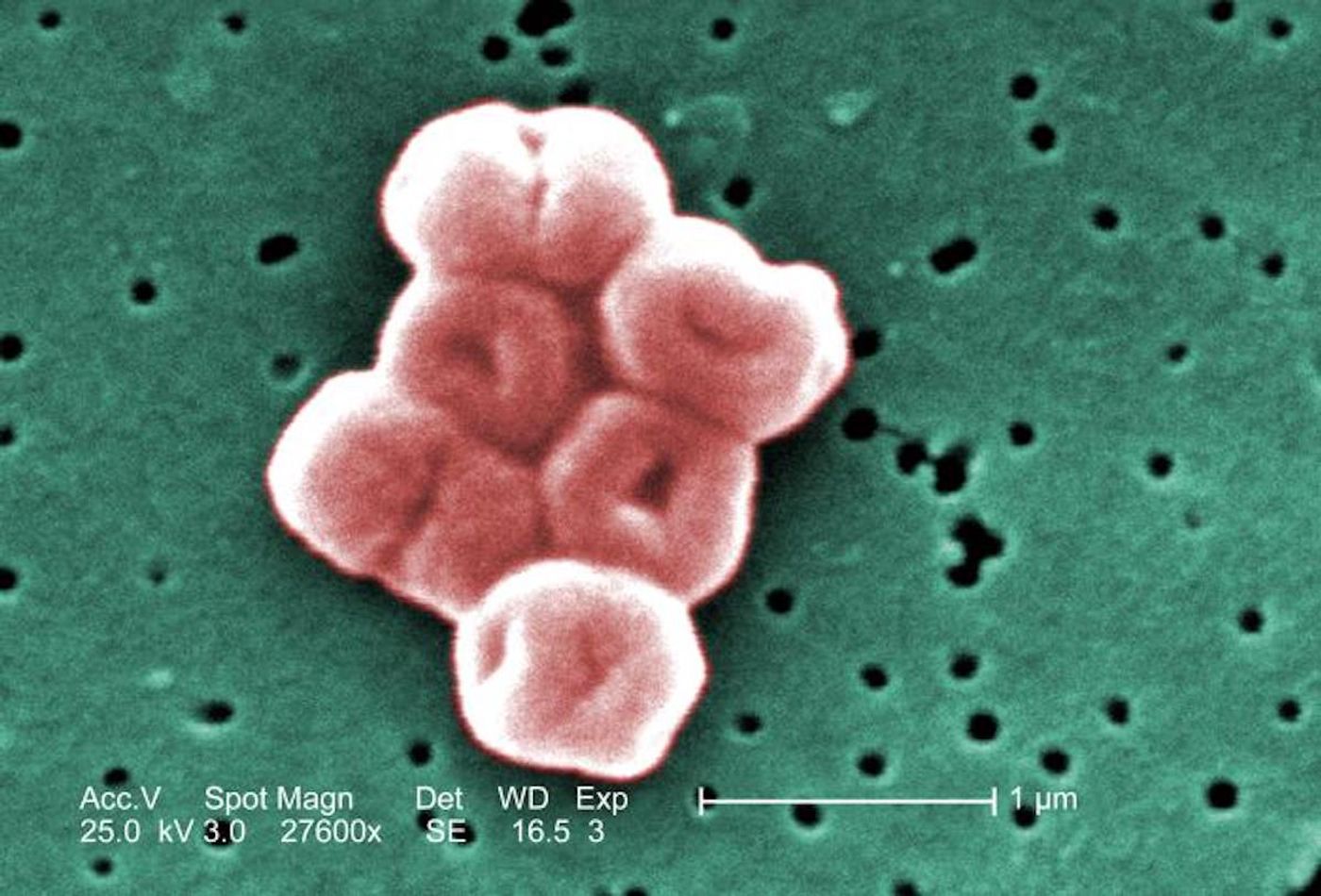
The bacterium Acinetobacter baumannii is harmless to most people. But it also causes many hospital-acquired infections in vulnerable people, and there are few antibiotics that can effectively eliminate the multi-drug resistant strains of this microbe. A. baumannii is known to be able to easily acquire drug-resistance. When critically ill patients become infected with A. baumannii, mortality rates can reach 20 to 60 percent.
The microbe is also tough. It resists disinfectants, and it easily survives when it dries out thoroughly. The pathogen can also infect people after it has dried, and can still cause serious cases of pneumonia. The World Health Organization has noted that there is an urgent need to develop antibiotics that can destroy A. baumannii.
Scientists have now studied A. baumannii, and how it can live through desiccation. Tests showed that the microbe could be reanimated even after it was dried for seven months. Strains of A. baumannii that have been recently isolated directly from clinics are also more resistant to drying than older strains; the microbe seems to be getting even hardier. In a mouse model, researchers found that A. baumannii infections were more serious after the microbe had dried out and been rehydrated.
Reporting in Cell Host & Microbe, a genetic analysis has now revealed that the bacterium carries proteins the researchers called "desiccation tolerance proteins," named DtpA and DtpB. The amino acid sequence of these proteins are unusual; they are intrinsically disordered proteins, which means they don't take on a three-dimensional structure that remains more or less the same, like most proteins. The DtpA and DtpB proteins are also hydrophilic; they don't like to mix with water.
Intrinsically disorder proteins have been found in other organisms like yeast and nematode worms, and some animals that are known to be unusually tenacious like tardigrades. DtpA and DtpB are some of the only intrinsically disordered proteins that have been found in bacteria, so far.
When the researchers put the DtpA gene from A. baumannii into another microbe, the recipient became more resistant to water loss. That characteristic could be very useful for probiotic bacterial strains that are meant to be used therapeutically, but would usually not survive desiccation. Heat-inactivated or dried bacteria or bacterial proteins might still be medically useful if they were bolstered with DtpA, for example.
"We think these proteins may have a valuable commercial application for preserving [the] activity of protein and probiotic therapeutics," said Eric Skaar, Ph.D., MPH, of Vanderbilt University Medical Center.
This study could also create effective strategies to eliminate A. baumannii from healthcare settings, where they do so much damage.
Sources: Vanderbilt University Medical Center, Cell Host & Microbe