Preserve scRNAseq Sample Integrity and Biological Diversity: Enabling QC Cost Savings
QC of samples is an integral step for ensuring reproducible, single-cell RNA sequencing data without compromising cell integrity, viability, or sample purity. Poor sample quality will end up costing labs money when sequencing by wasting reads on dying cells. Harsh prep or storage kills cells leading to incomplete biological diversity within a sample.
The HIVE™ scRNAseq Solution’s gentle isolation of single cells enables researchers to capture biological diversity, even when working with fragile cells, including cells lost by other methods. The HIVE™ scRNAseq Solution is an innovative technology developed by Honeycomb Biotechnologies which integrates sample storage and single cell transcriptional profiling into a complete workflow, solving the issues limiting single cell RNA analysis. It enables capture, storage, and easy processing. QC of samples prior to loading on the HIVE™ scRNAseq Solution, or any other scRNAseq device, helps labs optimize costs and overcome poor quality data.
Trypan blue staining, brightfield microscopy, and fluorescent cell counting can all be used to measure the viability and purity of a sample. Of these QC methods, research indicates fluorescent cell counting measures viability and purity more reliably either trypan blue staining1,2 or brightfield microscopy3. Brightfield microscopy can misidentify contaminating red blood cells (RBCs) and trypan blue staining can overestimate viability. Both of these QC methods can lead to overestimating cell counts which in turn leads to wasted reads when sequencing increasing costs.
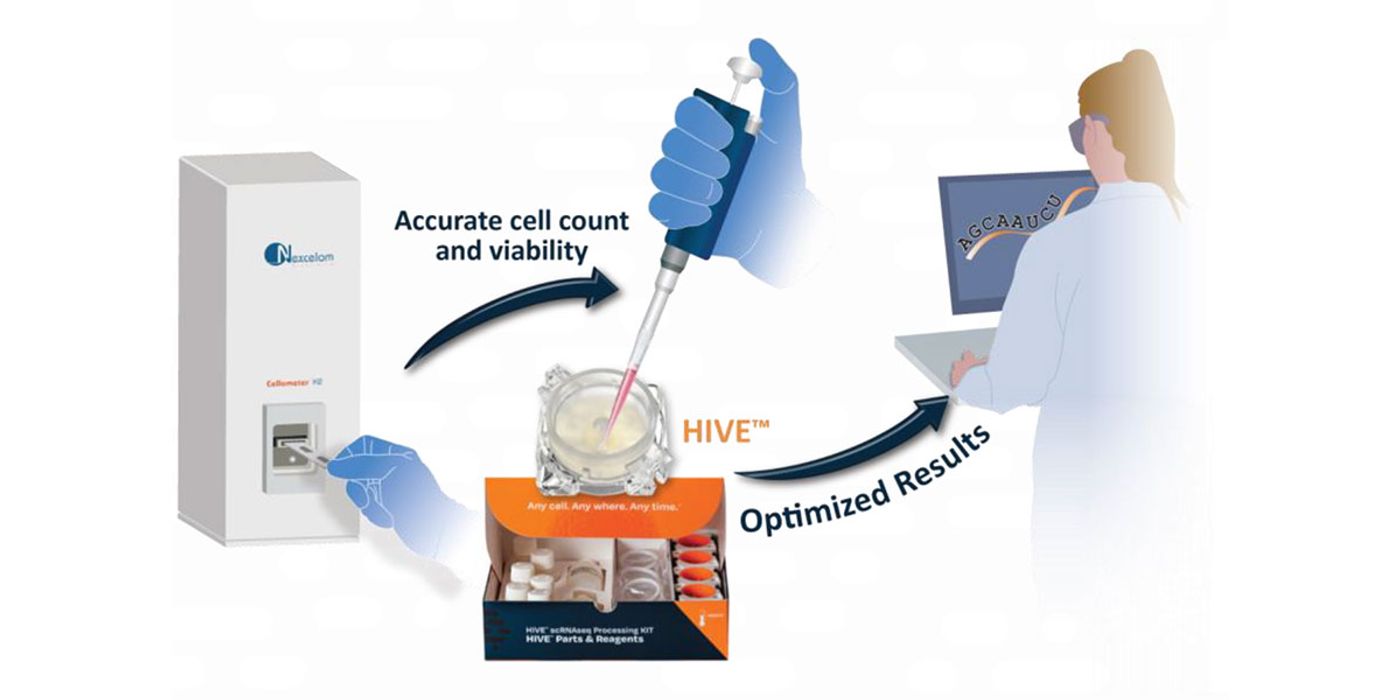
Leveraging the Cellometer® K2 Cell Counter to count cells prior to loading on the HIVE™ scRNAseq Solution, optimizes costs, and helps labs avoid issues arising from poor sample quality, such as sample bias, low cell recovery, low gene counts, and high mitochondrial reads. QC using the Cellometer® K2 Cell Counter allows labs to rapidly optimize sample prep and storage conditions and to adjust sequencing for optimal reads per cell.
The Cellometer® Device Automatically Measures Cell Counts, Viability, & Purity:

Combining the HIVE™ scRNAseq Solution which enables gentle collection and storage of single cells along with the Cellometer® K2 Cell Counter to ensure proper cell loading of the HIVE™ device, saves sequencing dollars while capturing the biological diversity of samples, even those samples containing fragile cells.
Learn more about the HIVE™ scRNAseq Solution
1. Chan, L., Rice, W., Qiu, J. Observation and quantification of the morphological effect of trypan blue rupturing dead or dying cells. PLoS One 15, 1 (2020). https://doi.org/10.1371/journal.pone.0227950
2. Chan, L., Kuksin, D., Laverty, D., Saldi, S., Qiu, J. Morphological observation and analysis using automated image cytometry for the comparison of trypan blue and fluorescence-based viability detection method. Cytotechnol 67, 3 (2015). https://doi.org/10.1007/s10616-014-9704-5
3. Chan, L., Laverty, D., Smith, T., Nejad, P., Hei, H., Gandhi, R., Kuksin, D., Qiu, J. Accurate measurement of peripheral blood mononuclear cell concentration using image cytometry to eliminate RBC-induced counting error. J Immunol Methods 388, 1-2 (2013). https://doi.org/10.1016/j.jim.2012.11.010
For research use only. Not for use in diagnostic procedures