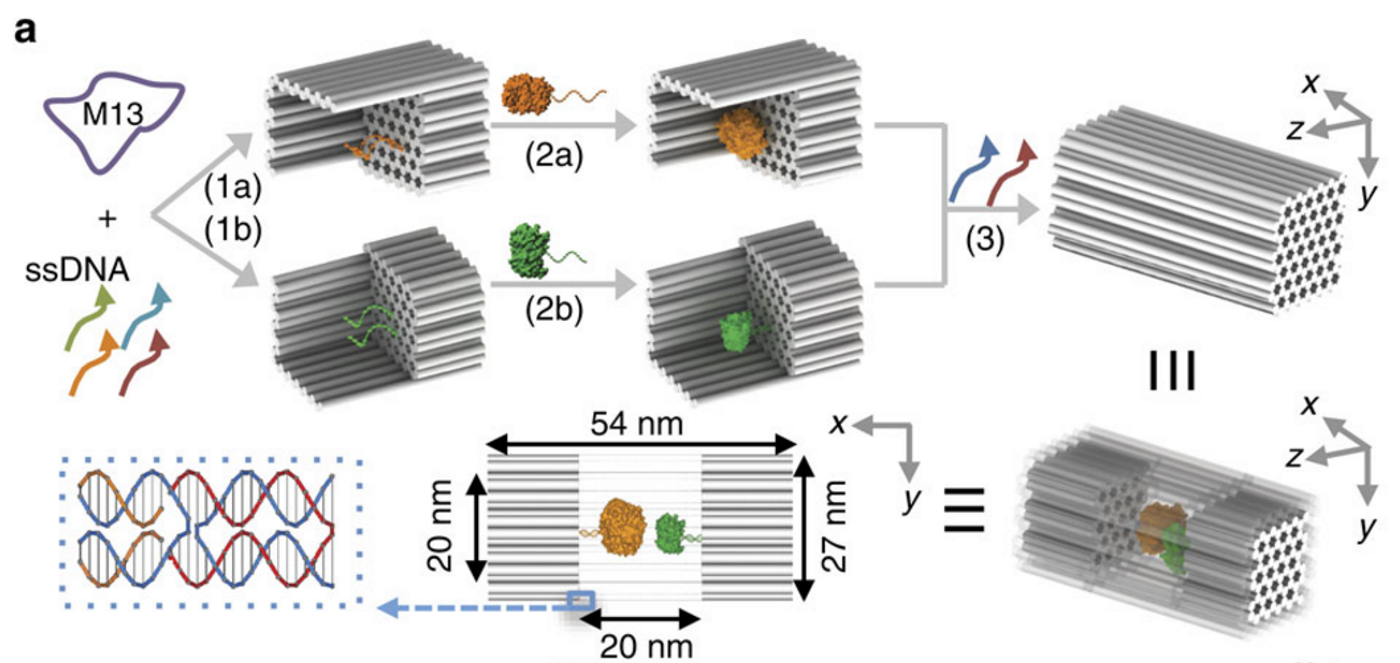
The cell is intelligently designed. Various membrane-bound organelles (mitochondria, lysosomes, etc) create their own perfect environment to run their enzymatic reactions. However, synthetic biologists have now been able to build their own compartments that can run enzymatic reactions even more efficiently than in the cell. A group led by Hao Yan out of Arizona State University has built DNA nanocages that house, and enhance, enzymatic reactions.
Other groups in the past have tried similar constructs using viral protein particles, but that is less desirable than using DNA for several reasons. First of all, the protein cages were not able to encompass large enzymes because of steric hindrance. Steric hindrance is a fancy way of saying that the amino acid side chains from the protein box got in the way of the side chains from the enzyme. Secondly, DNA is a better building material due to its “programmable, sequence-driven self-assembly.” The complementary base pairing that is intrinsic to DNA allows it to self-assemble into 3D structures. In nature, that means the double helix that we all know and love, but scientists can manipulate those properties to make DNA create a 54 nanometer box.
To run the enzymatic reactions, the boxes are first assembled as two half-boxes, with one half being loaded with the enzyme and the other half with the substrate. The two halves then associate, bringing the enzyme and substrate together (
video). The DNA nanocages also have small pores that allow cofactors for enzymatic reactions to diffuse through, such hydrogen ions. Large molecules cannot diffuse through the box, and the enzyme and substrate within are protected from degradation.
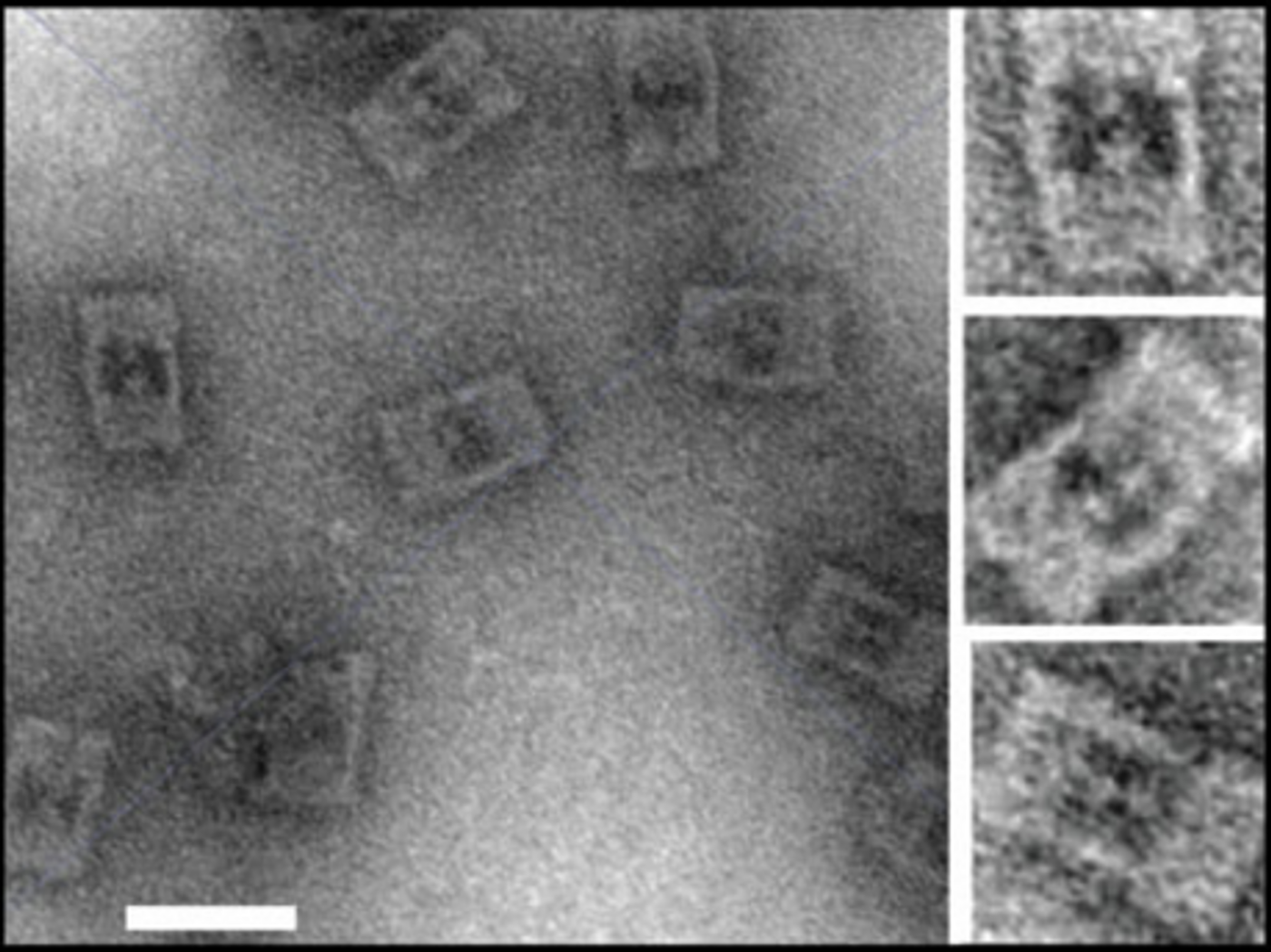
Overall, the nanocages increased enzymatic activity between 4-10 fold and increased the turnover rate (number of substrate molecules converted per second).You might think that the enhanced enzymatic activity is simply because the DNA nanocages are bringing the enzyme and substrate close together, but that is actually not the case. The majority of the effect the nanocages have on enzyme activity has to do with the charge distribution of the DNA. DNA carries a negative charge due to the phosphate groups, and in the highly structured nanocages, those negative charges hydrogen bond with water to created more stabilized layers of water within the cages. It is thought that the more structured water helps to stabilize the enzymes in their active conformations.
If you’re like me, you might read all this and think, “Super cool, but why?” This kind of synthetic biology actually has a lot of translational appeal. DNA nanocages could be used to create more precise enzymatic environments in smart materials, to make a very precise and targeted drug delivery system, to make industrial reactions more efficient, and to improve medical diagnostics. Additionally, the researchers point out that their findings can be used to increase understanding of cellular organization and why certain reactions are run in particular environments. Really, creativity is the limit for these kinds of advancements.
Sources:
EurekAlert and
Nature Communications