Zika virus has appeared from relative obscurity to cause big epidemics in South and Central America as well as Micronesia and French Polynesia. In most cases infection with Zika virus results in a fever associated with rash and conjunctivitis that resolves on its own. However, serious neurological phenotypes can also occur, including brain inflammation (meningoencephalitis) or Guillain-Barre syndrome.

Infection in pregnant women is a major concern, as Zika has been linked to catastrophic fetal abnormalities like microcephaly, intrauterine growth restriction (IUGR) due to placental insufficiency and spontaneous abortion. Because of this major public health concern, it is imperative to establish animal models of intrauterine Zika infection that will elucidate mechanisms of fetal transmission and expedite testing of treatments and vaccines. In addition, an in utero animal model of Zika infection will both establish causality and demonstrate clear proof that Zika is responsible for congenital defects seen in newborns.
Preliminary data suggest that Zika virus induced fetal abnormalities can occur in every trimester of pregnancy but the worst manifestations are associated with infections that occur in the first and second trimesters.
It was
reported in Cell that a group at Washington University School of Medicine, Saint Louis that had previously developed an adult mouse model of Zika virus infection has now created a mouse model of in utero transmission of Zika. They used their previous adult model, mice that don’t produce or respond to Type I interferon (IFN) - a large group of proteins that are critical for immune system regulation. While adult wild-type (WT) mice did not develop illness after they were infected with a Zika, the mice that lacked the IFN response did develop severe neurological disease, associated with high viral loads in the spinal cord and brain, and also experienced high rates of death.
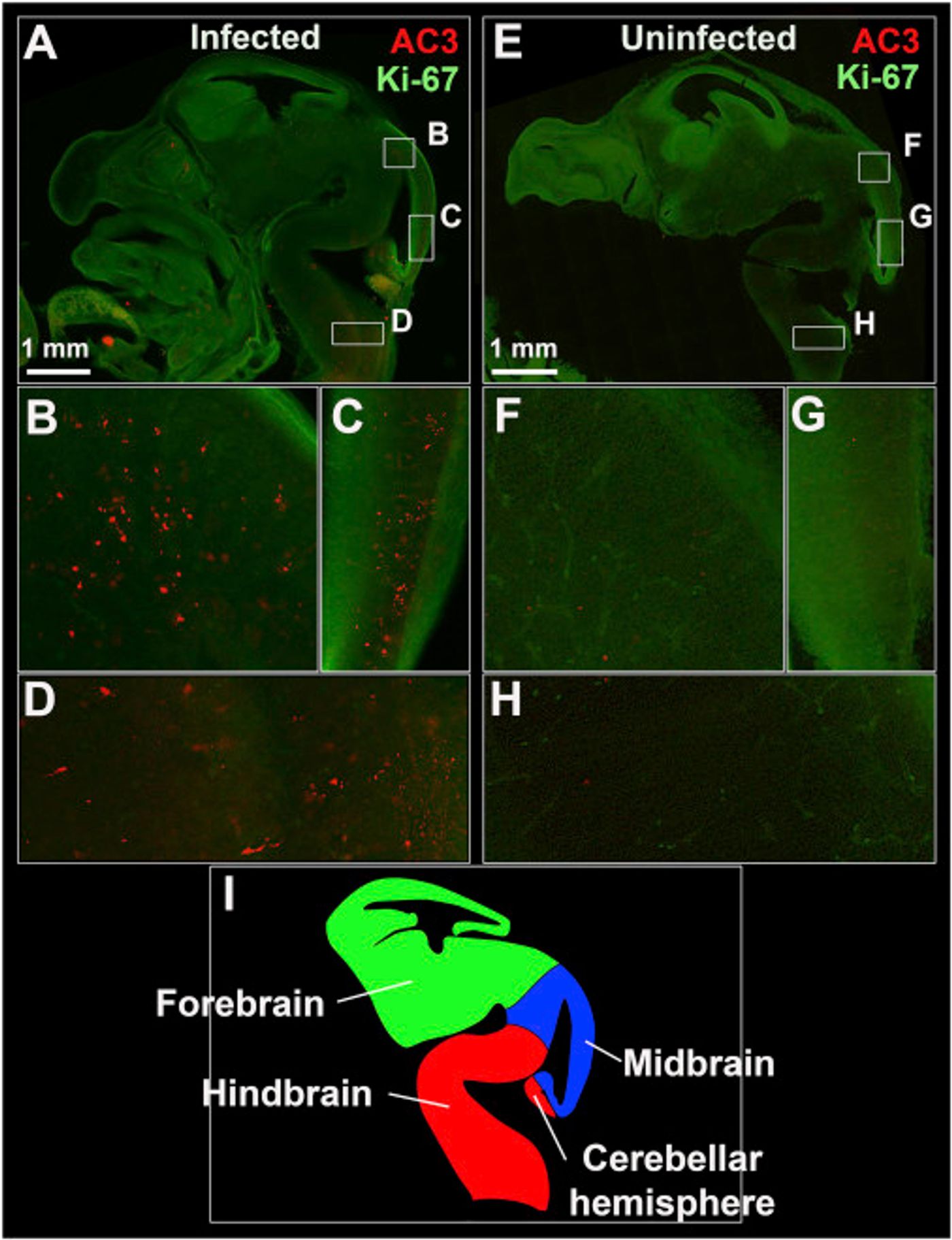
The researchers bred female mice without IFN response to WT males. It was found that Zika virus infected these pregnant mice and their placenta. This resulted in damage to the placental barrier, infection of the developing fetus, placental insufficiency and IUGR. In the most serious cases, Zika infection killed the fetus. When the pregnant mice received treatment in the form of an antibody that would block the IFN effect, infection did still occur but the consequences for the fetus were far less severe.
Their findings could establish reliable models for studying the mechanisms of in utero transmission of Zika, as well as testing of candidate therapies to prevent congenital malformations. The scientists also highlight the concern that Zika infection could happen in the fetus of an otherwise healthy looking parent, with unknown neurodevelopmental consequences. Animal models of Zika virus infection during pregnancy may also provide fundamental understanding of how the placental barrier can protect the developing fetus from viral infection, and why that protection fails in the context of some pathogens.
Source:
Cell