The US government panel that evaluates gene therapy trials, the National Institute of Health’s Recombinant DNA Research Advisory Committee (RAC), has approved a proposal by the University of Pennsylvania (UPenn) to test the CRISPR/Cas9 genome editing technology in a human patient. While there are still some other regulatory hurdles, this was a crucial and encouraging first step. Below, the video displays the CRISPR method.
While gene therapy has been attempted and has even found success in treating people before, this is the first use of the popular CRISPR tool to alter three different genes simultaneously for use in human patients, an exceptional achievement. The source of the funding for the trial, former Facebook president Sean Parker’s new $250 million Parker Institute for Cancer Immunotherapy, has also attracted a lot of attention to the study. While it is still unclear who might own the intellectual property surrounding this venture, Parker’s foundation has said that it will retain control of any patents on research it funds.
Eighteen people with a few types of cancers that are no longer responding to any current treatments will participate in the trial. The scientists plan to remove T cells, a type of white blood cell, from the patients and perform three CRISPR edits on them. First will be a gene insertion that causes T cells to target cancer cells. A benign virus will give the T cells a receptor for a protein, NY-ESO-1, usually present on tumor cells. When the T cells are reinfused into the patient, they should then attack tumors showing that NY-ESO-1 protein. This part of the plan has already been tested in people with multiple myeloma. While most tumors did initially shrink, the edited T cells eventually lost effectiveness and stopped proliferating.
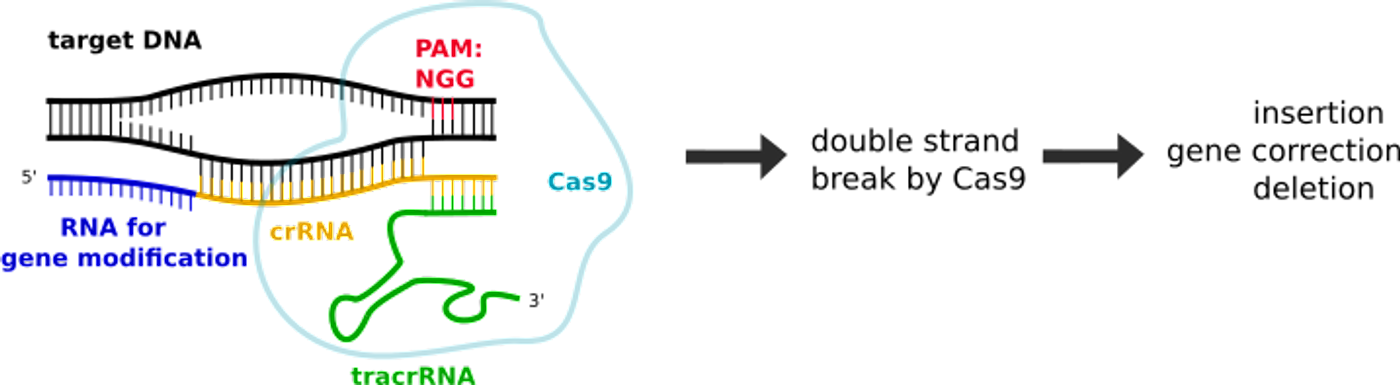
To hopefully address these issues, there will be additional uses of CRISPR on the T cells. Second, an edit to remove a natural T cell protein that could cause interference with this process. A third edit serves a defensive purpose; it removes a gene that identifies T cells as immune cells and prevents cancer cells from disabling them. After the successful edits, the cells will be put back into the corresponding patient.
“It’s an important new approach. We’re going to learn a lot from this. And hopefully it will form the basis of new types of therapy,” says one of three RAC members who reviewed the protocol, clinical oncologist Michael Atkins of Georgetown University.
UPenn will have to overcome an unfortunate history with gene therapy. In 1999 a young man in a trial there, Jessie Gelsinger, died participating. “Penn has a very extensive conflict and has a history,” says a bioethicist at Northwestern University, Laurie Zoloth.
One major concern of the committee arose from the financial interest UPenn has in the trial. Carl June of UPenn is the trial’s scientific adviser, and he holds several patents on gene therapy as well as having connections to Novartis pharmaceutical company. The RAC made several suggestions for avoiding such conflicts, and June claimed the university was taking care to manage such issues. RAC members have said they are being very careful with this proposal.
June thinks one of the biggest obstacles to success will be the potential for 'off-target effects' or the unintended editing of genes that are not targeted in the study. It is also possible that the immune system will mount an attack on the edited cells.
Mildred Cho, a bioethicist at Stanford University in California and an RAC member, thinks the time is right to move forward. “Often we have to take the leap of faith.”
“CRISPR technology provides an opportunity to profoundly manipulate cells,” according to a statement provided by Jenifer Haslip, a spokesperson for the Parker’s nonprofit foundation. “We’re excited to be part of the first clinical effort in the United States to combine these two powerful therapeutic approaches to treat a devastating disease like cancer."
The structure of the Cas9 protein is seen in the video above.
Sources:
Science ScienceInsider,
Nature News,
Parker.org,
MIT Technology Review