Intermediate filaments (IFs) are critical parts of the structure of cells. They are essential for both normal tissue structure and function. Enabling cells to withstand the stretch and pressure on the tissues in which they reside, they protect against problems like blistering and cataracts. Found in the nucleus (lamins) and the cytoplasm (cytoplasmic intermediate filaments), they take their name from their filament diameter.
Strangely, they have not been found in insects, and it was thought that an unknown group of proteins had taken on this function in those organisms.
Publishing in Cell Reports, new
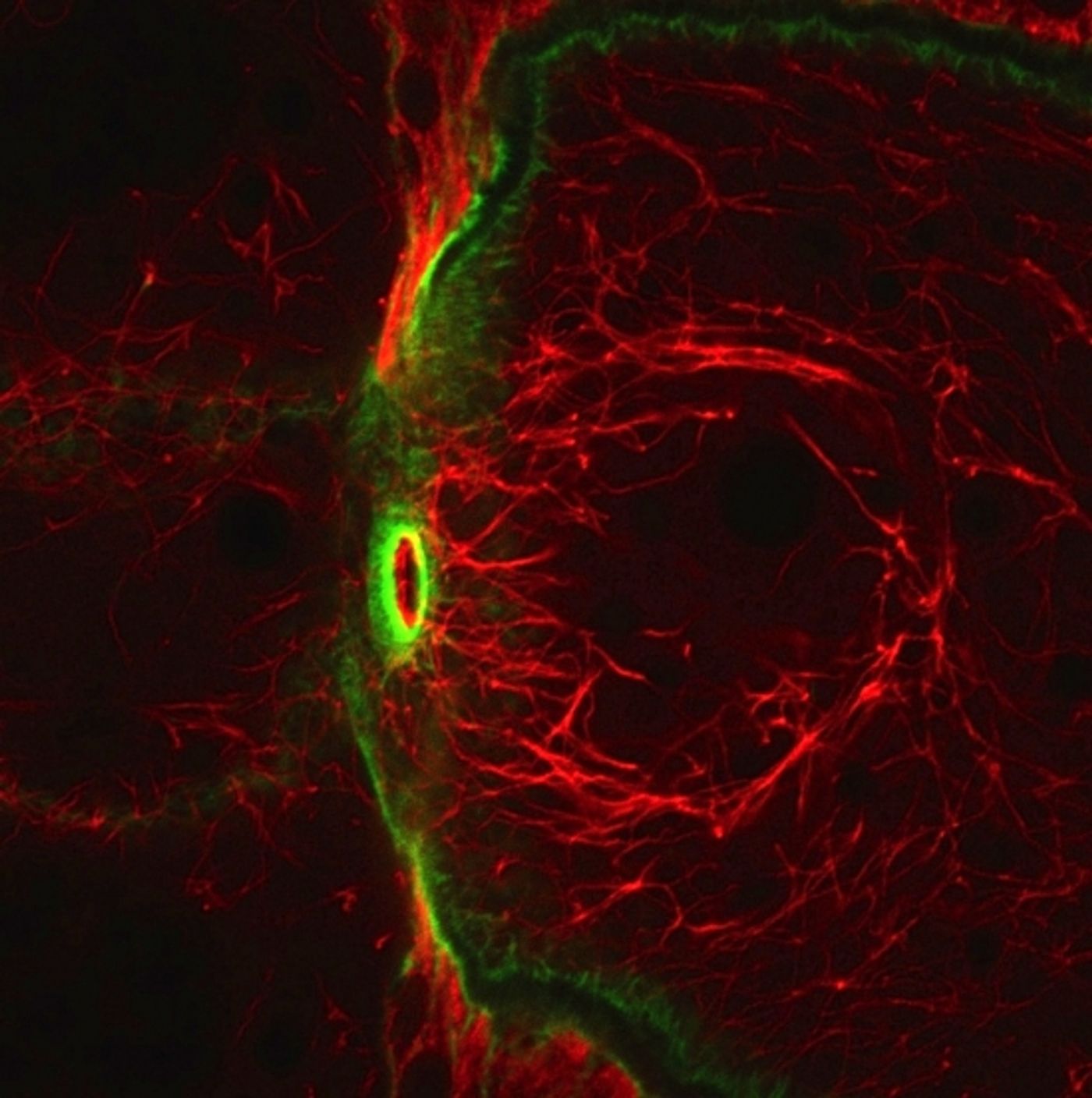
work conducted by biologist Denise Montell and her team of researchers at the University of California Santa Barbara (UCSB) may have the answer. They have discovered a protein that has IF-like characteristics in fruit flies. They observed that this unusual form of a protein called tropomyosin was in every cell type they analyzed.
"Believe it or not, fruit flies and humans have a lot of proteins in common, including conventional tropomyosin," explains Montell, the Robert and Patricia Duggan Chair of Mathematical, Life, and Physical Sciences in UCSB's Department of Molecular, Cellular, and Developmental Biology.
"Evolution started with tropomyosin and morphed it into a brand new protein with very different characteristics simply by adding a few bits to each end," she continues. "Like IFs -- but unlike normal tropomyosin -- this new protein makes filaments that are 'intermediate' in size. This means they are larger than F-actin filaments and smaller than microtubules, two other important structural filaments in cells." This newly discovered type of filament-forming protein may be analogous to some intermediate filaments in flies.
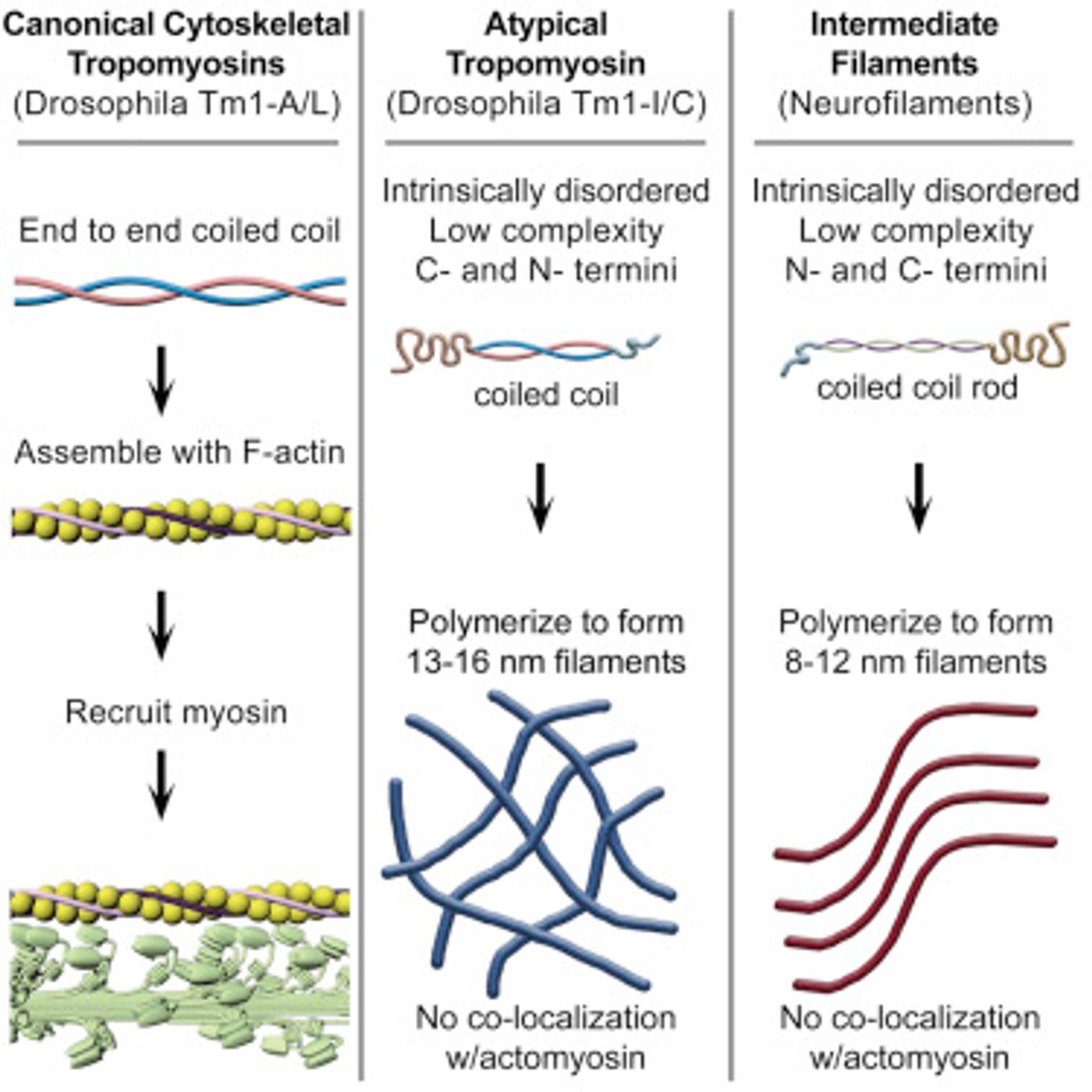
Normal tropomyosin creates a rope-like structure, called coiled coil. This thin tropomyosin coil normally winds around F-actin filaments. However, this newly discovered version doesn't resemble or behave like a normal tropomyosin and is coiled in only one small region. "We knew what it wasn't, but we wanted to know what it was," says Montell.
The team utilized a computer program which examined the uncoiled, disordered regions at each end of the protein to better understand the function of this unique molecule.
"While this configuration -- coiled coil with intrinsically disordered domains at each end -- resembled IF proteins generally, the novel tropomyosin differed from IFs in the details," Montell explains. "Still, we wondered whether this atypical protein might have filament-making capabilities or the ability to perform IF-like functions."
A colleague at the University of Texas Southwestern Medical Center, Masato Kato, made the protein in a tube to investigate conditions such as salt concentration and pH that are appropriate for different types of IF proteins. He determined that the environment in which this new protein from flies formed 13- to 16-nanometer-wide filaments had never previously been described.
"We found all kinds of similarities in biochemical behavior between this protein and IF proteins despite the differences in sequence," Montell says. "Because our protein seems to have some properties in common with IFs, it may be the answer to the long-standing question of how flies survive without actual IF proteins. But like any exciting discovery, it also raises many more questions."
Sources:
AAAS/Eurekalert! via
UCSB,
Cell Reports