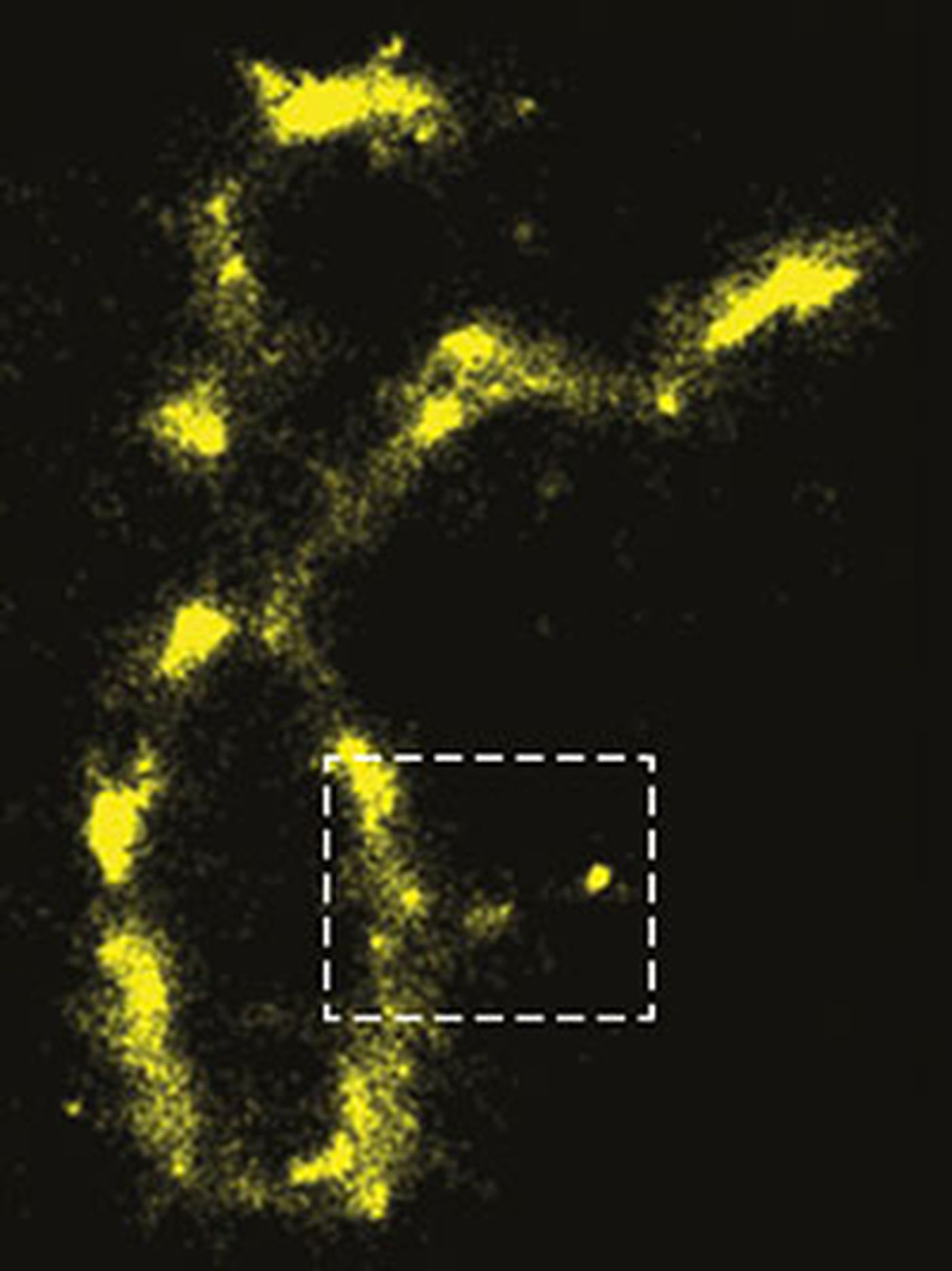
It’s long been accepted that proteins, RNA and DNA have to be stained to be visualized under fluorescence; the belief has been that macromolecules in living cells don’t fluoresce by themselves. A research team at Northwestern University in Illinois has found however, that they do naturally fluoresce after all. The findings could greatly improve imaging and molecular studies at the micro- and nano-scale.
"There are textbooks that say biological molecules don't absorb light and don't fluoresce," said Zhang, an Associate Professor of Biological Engineering at Northwestern University. "It's what everyone learns; it's a part of training, so nobody questions it."
"Everybody has overlooked this effect because nobody asked the right question," commented Vadim Backman, the Walter Dill Scott Professor of Biomedical Engineering at Northwestern's McCormick School of Engineering. "It sounds cliché, but you get the answer to the question you ask. When we actually asked the right question, we got a very different answer than expected."
The researchers,
publishing in the Proceedings of the National Academy of Sciences, figured out that when cellular molecules are illuminated with visible light, they are excited such that they can be imaged without having to use fluorescent staining. They determined that when using the right wavelength, they fluoresce more powerfully than they do when stained.
So why hasn’t anyone observed this phenomenon before? The scientists say the molecules were only in a “dark state” - meaning they were not emitting or absorbing light. However, that apparently does not mean they remain in that state forever.
Backman uses athletic interval training as an illustrative example. "Sprinters alternate running very, very fast and resting," he explained. "You might catch them when they are resting and assume they aren't doing anything. That's what DNA and proteins do. They fluoresce for a very short time and then rest for a very long time."
Zhang highlights how advantageous it would be to eliminate the staining process. Such labeling is destructive to cells; after the dye is applied to living cells, they die. Stains specially created for use in living cells can only make the cells die more slowly. "This is ideal because staining is toxic," said Zhang, "and it makes imaging less precise."
"The cell might die in two hours, so you can still do imaging in the first half hour," Backman continued. "But what exactly are you measuring? What are you actually seeing? Are you looking at real processes of the cell? Or are you looking at processes in a cell that is about to die? Nobody knows."
Researchers are sure to follow up on this exciting finding, and hopefully we’ll be hearing more about this soon; not only the replication of the finding but its application to data acquisition. It will be very interesting indeed to compare results obtained without staining to what was seen by with it.
Sources:
AAAS/Eurekalert! via
Northwestern University,
PNAS