New Research May Change Accepted View of Cell Cycle Control
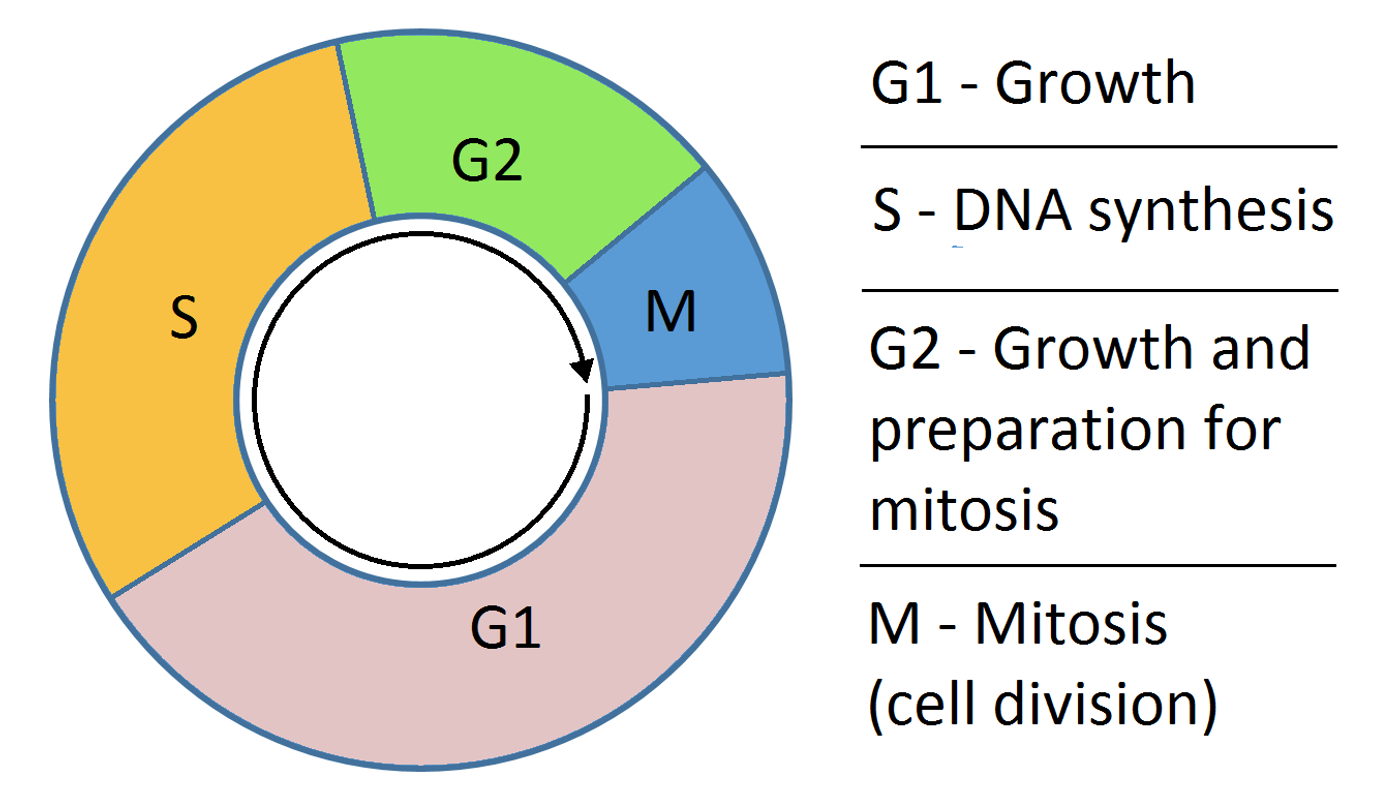
The cells of organisms move through growth stages, duplicating DNA and eventually dividing, and control of that cycle was thought to be understood. The 2001 Nobel Prize in Physiology or Medicine was awarded for the demonstration that the cyclin-dependent kinase complex regulates the cell cycle. New work however, has followed up on some of the unexplained questions remaining in cell cycle control; researchers have discovered that a metabolic oscillator acts as the conductor of cell division.
Some data has stood at odds with the accepted view; cells can divide in the absence of the cyclin machinery, for one. For another, in cells where the cycle is arrested, proteins of the late cell-cycle keep on oscillating. "But there were signs that [we didn't have] the complete story," said University of Groningen system biologist Matthias Heinemann. "We knew that metabolism often oscillated in synchrony with the cell cycle. So maybe, this was an autonomous control mechanism."
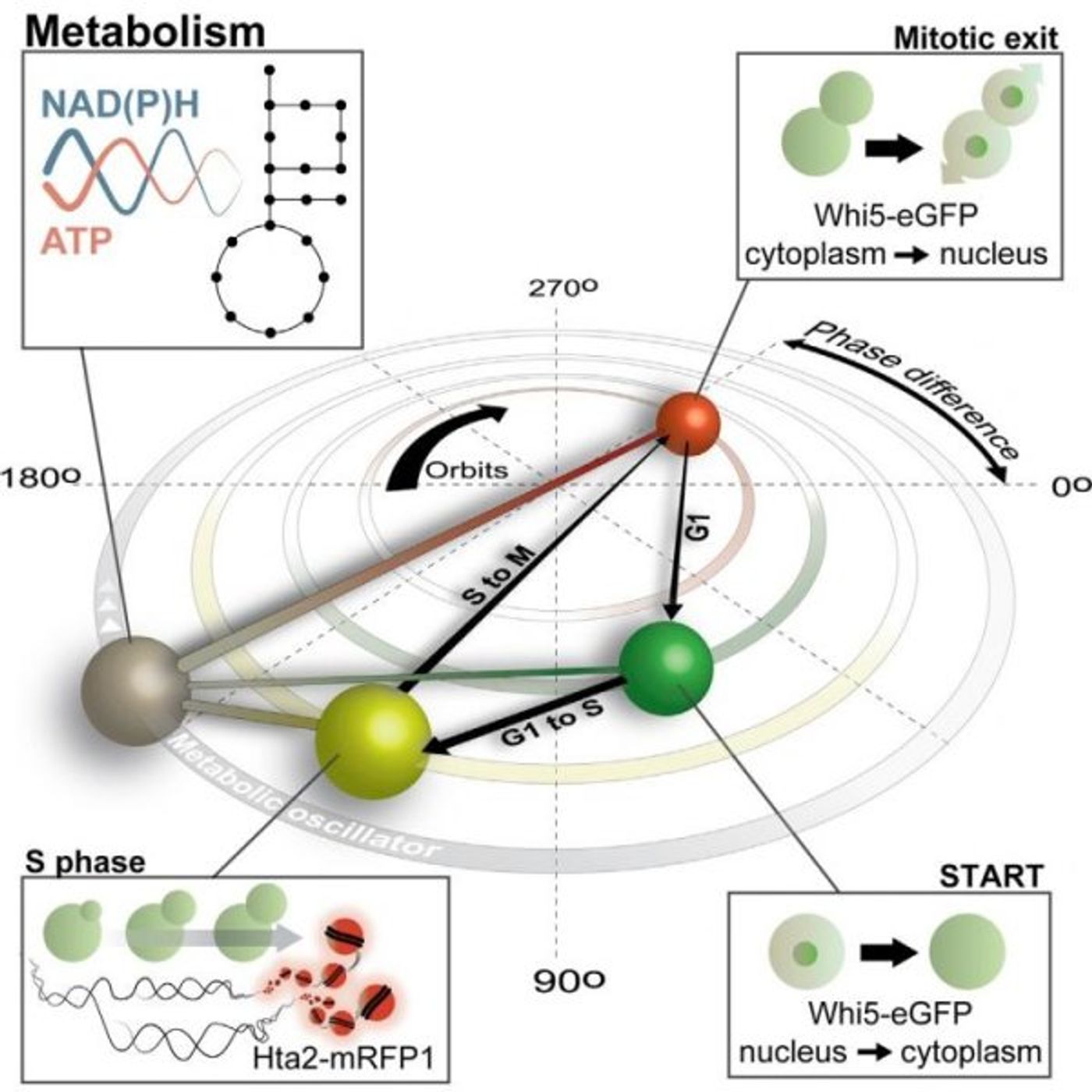
Heinemann looked at yeast cells growing in microfluidic channels so individual cells could be observed under a microscope for several days. The addition of fluorescent markers enabled the measurement of metabolic markers NADH, an electron carrier, and ATP, an energy source. From that, changes in the levels of those molecules that indicated clear oscillations were seen, usually moving along with the cell cycle. Heinemann added, "But we also noticed that occasionally cells did not divide, and that these cells still showed metabolic oscillations."
Metabolism appeared to be oscillating independently of the cell cycle; well fed cells had rapid oscillations, poorly fed cells oscillated slowly. "We argue that metabolism and the cyclin-dependent kinase complex are coupled oscillators, which together orchestrate the growth and division of eukaryotic cells, but when metabolism is slowed down or sped up too much, the cell cycle can't keep up and stops," said Heinemann. The cyclin-dependent kinase complex moves to the rhythm of the metabolic oscillations, until it can't keep up. The graphical abstract from their work, published in Molecular Cell, is shown above.
The researchers suggest a system where the metabolic oscillations push the cyclin-dependent kinase complex through the cycle, controlling the timing of the various cell cycle stages. This new work could change a lot of textbooks.
"The current view is too narrow and cannot explain why cells still divide when part of the cyclin-dependent kinase complex is removed," commented Heinemann. It is logical that evolution would favor a focus on the workings of metabolism. "You would expect the earliest cells or proto-cells to have a simple control system to regulate division, and metabolism would be the obvious candidate." This new work could influence clinical research as well. "Most tumor cells have a very high metabolism. Interfering with metabolic processes could be a way to stop them from proliferating."
If you'd like to know more about gene oscillation networks, watch the following MIT lecture.
Sources: Science Daily via University of Groningen, Nature, Molecular Cell