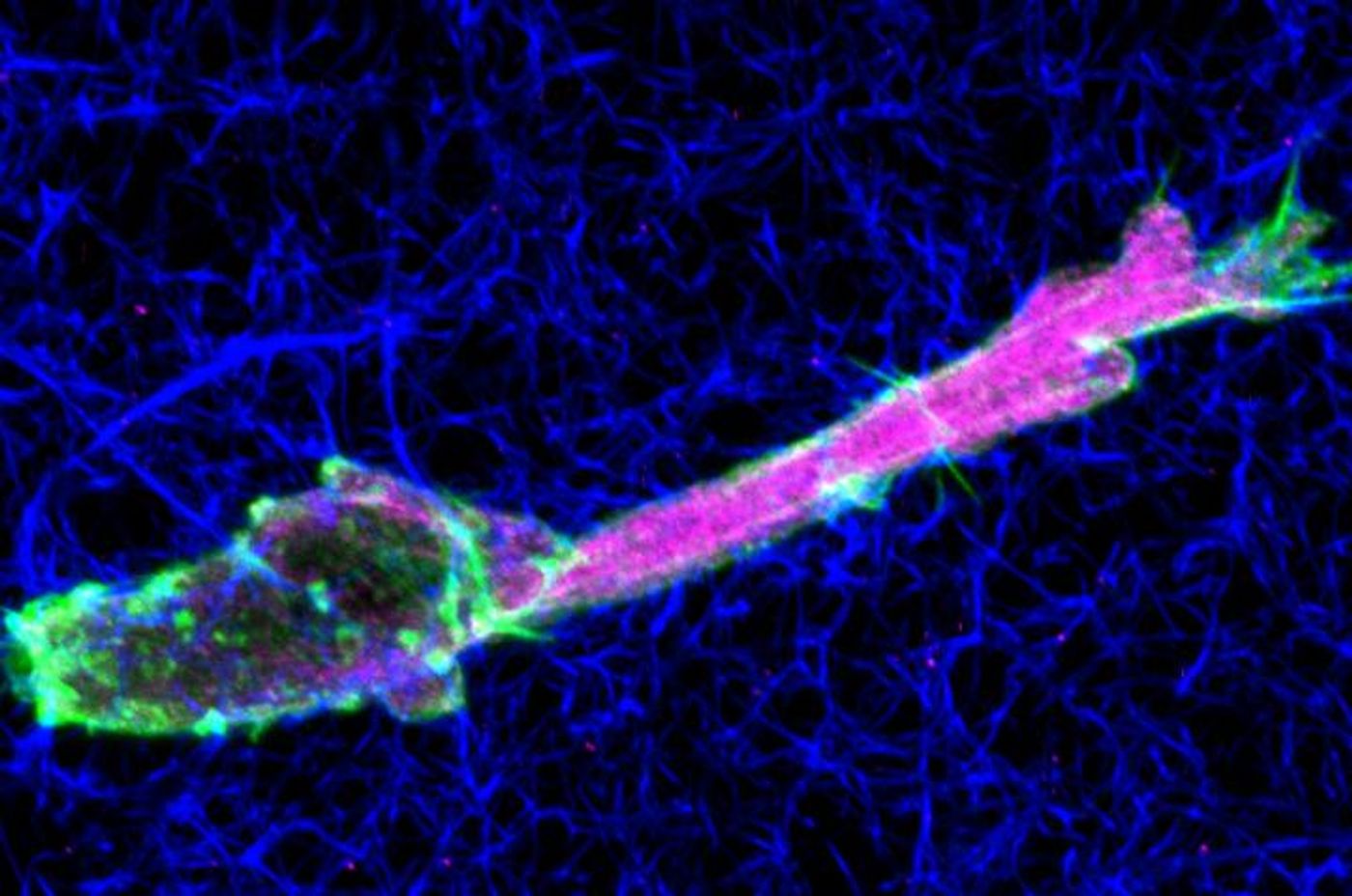
Tumor Cells Move Through Tissue in a Unique Way
The movement of tumor cells is dangerous to human health; metastatic cancer can migrate to adjacent tissue and even other parts of the body, making it deadly. Containing cancer to one area could make it easier to treat, reducing the risks it presents and improving outcomes for patients. "Therapeutically preventing the inappropriate movement of metastatic tumor cells could be used in combination with existing chemotherapies to increase patient survival," said Ryan Petrie, an Assistant Professor in Drexel's College of Arts and Sciences.
Petrie’s research team has investigated the movement of cancerous cells, specifically fibrosarcomas, and discovered that they cannot move in the way that normal cells do when they squeeze through constricted parts of three-dimensional spaces. The nuclei of cells are large and rigid and can’t fit through a tight matrix easily; when normal, healthy cells are faced with such an obstruction, they alternate to another mode of motion. They can move their nucleus through the body of the cell like a piston moving in an engine. Petrie’s team has determined however, that fibrosarcoma cells are unable to move their nucleus like that in the presence of highly active, specific protease enzymes. The videos below show experimental confirmation that the protease inhibition drives the piston-like movement. From their report: "In control cells moving through 3D CDM [matrix], the nucleus and trailing edge tended to move simultaneously. Upon protease inhibition, however, the nucleus and trailing edge moved independently of one another, consistent with the nucleus being pulled forward by the nuclear piston mechanism.."
The tumor cells resort to another method to get past hurdles. Instead of powering through with a motor without damaging surrounding structures, tumor cells simply tear through the matrix, using molecules called matrix metallooproteinases to enlarge openings and give themselves room to maneuver. The new work, using cattle skin collagen and rat tails to reveal these details, was published in the Journal of Cell Biology.
"Cell migration is a lethal characteristic of metastatic tumors, where malignant cells begin to move inappropriately and spread through the body to form secondary tumors," Petrie commented. "To fully understand the mechanisms which drive normal and pathological cell movement, we must study cell migration in three-dimensional environments, such as the ones found in our tissues."
This new data is not only set to inform cancer research, but could inform wound healing therapeutics. "Promoting movement of fibroblasts in specific three-dimensional tissues like dermis [skin] and cartilage could help to heal difficult-to-treat wounds," explained Petrie. "Understanding the fundamental molecular mechanisms driving the movement of these cell types will be essential for designing rational therapeutic strategies in the future."
Understanding the various modes of migration in both normal and cancerous cells is important, and there is more work to be done in order to determine how the variations arise.
"The next step will be to untangle the intracellular signaling pathways which dictate cell behavior to understand precisely why tumor and normal cells move differently in the same three-dimensional environment," Petrie said. "We speculate these signaling pathways will provide the best candidates for drugs aimed at promoting or reducing cell movement in the future."
Sources: AAAS/Eurekalert! via Drexel News, Journal of Cell Biology