Structural Study of Multidrug Resistant Pump Sheds Light on Mechanism
Cells are actually able to get around the effects of medication by kicking it out; cancer cells are especially known for this striking behavior, with chemotherapy drugs simply removed from the cells they're meant to annihilate. Scientists have gained new insight into the mechanisms behind the transporter responsible by figuring out its structure at the atomic level. This work, by researchers at Rockefeller University, has been published in Cell. This work could lead to improved treatments for cancer, ones that aren't subject to elimination by the pump. The video below features the pump.
"This molecular machine ejects numerous anticancer agents, as well as other drugs. However, no one understood how it can recognize and remove such an impressive variety of substances," explained the leader of the research, Jue Chen, the William E. Ford Professor and head of the Laboratory of Membrane Biology and Biophysics. "By examining how this drug resistant pump binds to its cargo before transporting it, we have found an answer," she added.
This pump, the multidrug resistance protein MRP1, was found in 1992 in lung cancer cells that were resistant to drugs. However, it's not only found on tumor cells. In the body it is normally also located on the barrier that shields the brain; that barrier can allow only very selective things through, but allows for many molecules like hormones and signaling chemicals, to be exported ad pass out of that barrier. There, the pump can not only get important stuff out, it sometimes also gets rid of chemicals we'd rather have in, like antidepressants and antibiotics.
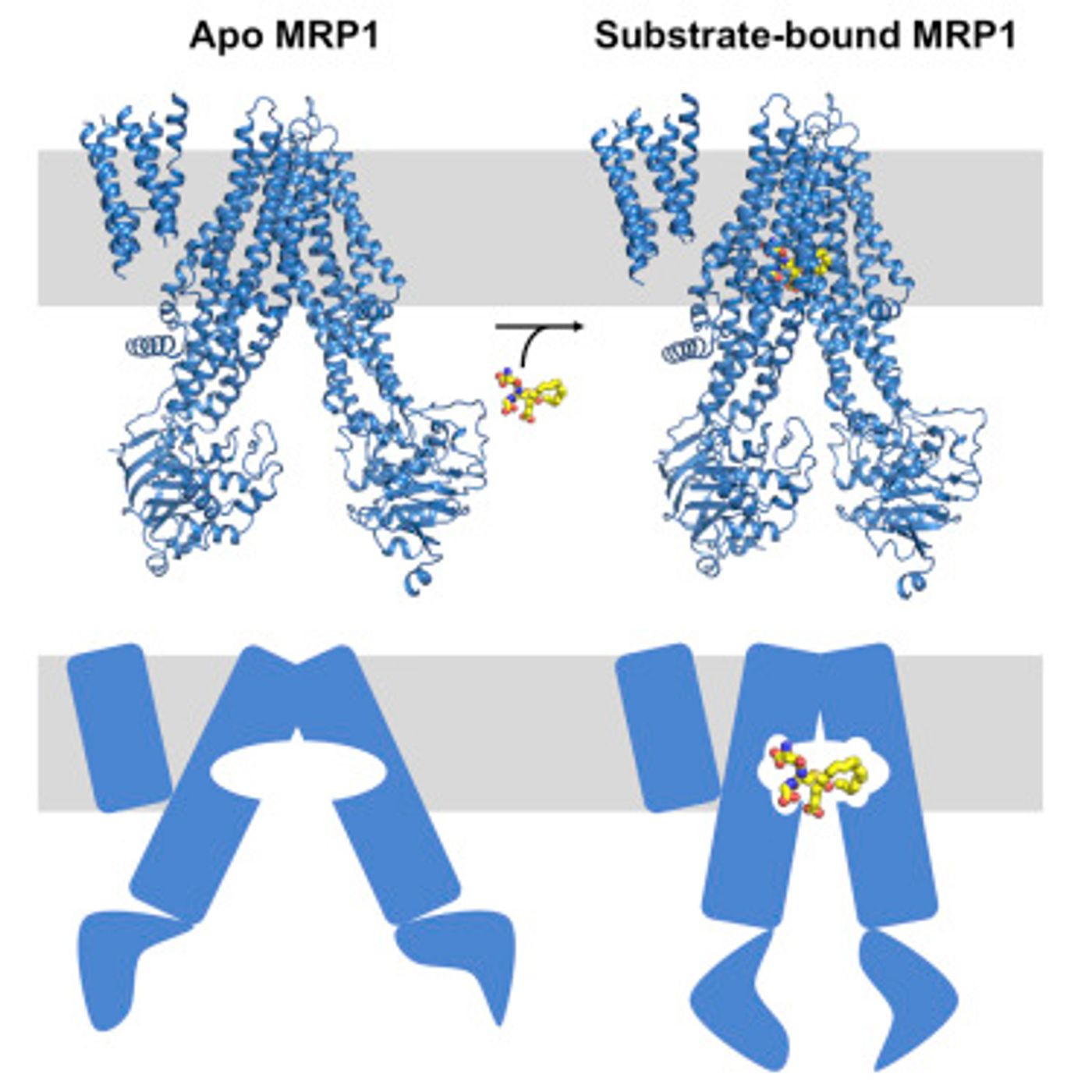
Membrane pumps and channels are usually very particular about what the move across the membrane. MRP1 doesn't work that way, but researchers haven't found an explanation for why that is, especially because the structure of MRP1 was not known. That is the usually starting point for learning about the cargo that a pump engages. Those studies are done by crystalizing the protein, but scientists weren't able to do that with MRP1.
Proteins charged with transportation across a cell's membrane tend to be picky, accepting only particular types of cargo, in some cases a single molecule. MRP1 is different, but because researchers couldn't get a detailed look at it, they couldn't be sure why. That's because the standard method for determining the structure of a molecule required them to turn it into crystals, something they couldn't do with MRP1.
For this work, Chen and her postdoc, Zachary Johnson, avoided that hurdle by utilizing electron cryomicroscopy, an imaging technique in which molecules in solution are frozen instead of being crystallized. Mark Ebrahim and Johanna Sotiris in Rockefeller's Evelyn Gruss Lipper Cryo-Electron Microscopy Resource Center assisted the project, enabling the capture of images of MRP1 on its won and while carrying its cargo.
Deep within MRP1, the researchers identified a special pocket that tucks cargo away; the pocket is a flexible structure composed of the sides of the pumps it can carry many different sizes of cargo. There are also two parts of the pump with their own chemical characteristics. Apparently many molecules transported by MRP1 match up with the two halves, while those that don't are attached to a molecule from inside the cell that acts as a filler.
After the cargo gets into the pocket, it connects the two parts of MRP1 that are then pulled together. Chen and Johnson observed this in action, like a clothespin being pinched shut.
The researchers hope their results will have implications beyond just MRP1; there are many members of this protein family, and others also eject drugs from cells."Ultimately, we hope these and other structures like them could serve as templates for designing chemotherapy drugs the pumps don't recognize, or inhibitors that block them," Johnson explained. "It's possible that such a strategy could also improve the effectiveness of other drugs, such as those that need to reach the brain."
In the following video, Dr. Chen presents a lecture at NIH on the maltose transport system, which is considered a model for the study of ABC transporters, one of which is MRP1.
Sources: AAAS/Eurkealert! via Rockefeller University, Cell