Revealing the Connections Between Organelles in Cells
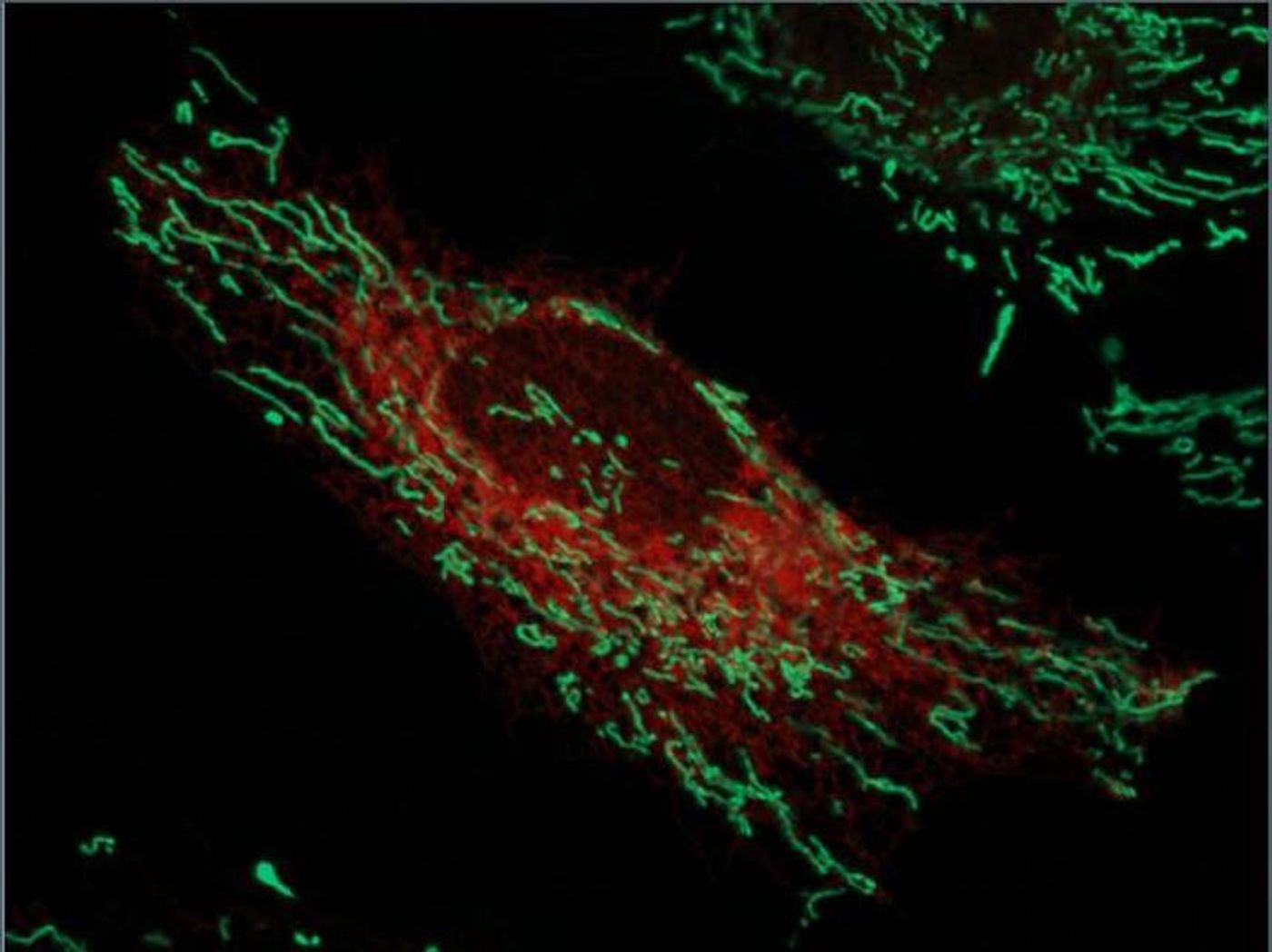
Our body is made up of many different kinds of cells, and inside each cell are organelles, tiny machines that help carry out cellular functions. New work reveals more about a network connecting two organelles. Reporting in the Journal of Biological Chemistry, this work shows how the endoplasmic reticulum (ER) and the mitochondria are joined in a way that allows for calcium and lipid exchange, signaling, and physiological control.
"Think of [an organelle] like a ferry docking at one site, unloading and loading passengers and cars, and then going to another site and doing the same thing," explained the investigator who oversaw the project, Jeffrey Golden, a Professor at Brigham and Women's Hospital and Harvard Medical School. "Their ability to dock, load, and unload cargo requires guides or ramps of specific width and heights that connect the boat and land or they cannot freely load and unload."
When the guides or ramps between the ER and mitochondria are dysfunctional, a neurodegenerative disease like Alzheimer's, Parkinson's and Huntington's disease can result. Although this system has been studied in yeast, not as much as known about how it works in people, and the scientists set out to learn more.
To that end, Golden's collaborator Ginam Cho and research fellow Il-Taeg Cho used a new labeling method. In it, an enzyme called APEX attaches biotin to proteins; for this work, the researchers engineered cells that make mitochondria with APEX attached. By the adding biotin to the cells, proteins near the mitochondria would be labeled. After isolating only the parts of cells that contained ER, proteins with biotin attached could be purified and identified.
Mass spectrometry was then used to identify the proteins that had been found in the ER. The use of APEX was meant to ensure that only proteins with biotin attached, and thus were close to the mitochondria, were isolated. The biotin thus showed that the proteins they found had been in contact with both organelles.
"It was previously feasible to only look at one molecule at a time to assess what it interacted with," said Golden. "The method we have used is more rapid and allows an unbiased look at a whole system and what's happening at that organelle's interface.”
This method led the researchers to an ER protein called RTN1a, which has previously been shown to influence the shape of the ER. The researchers confirmed this protein can help mitochondria attach to the ER as well. This work suggests that RTN1a defects might affect neurodegenerative diseases, but the data must first be confirmed in neurons. Golden thinks that proteins that affect ER-mitochondrial contact might not be the same in all cells.
"Does the liver use the same proteins to control these kinds of interactions that neural cells do? Is one more important for calcium exchange and another set of proteins more important for lipid exchange?" queried Golden. "I think there's a lot of cell biology that we just don't know and could be answered [with this method]."
The investigators are now using the APEX-mass spectrometry protocol to look at how proteins involved in ER-mitochondrial contacts differ in cells derived from healthy and patient neural cells.
"There are a lot of interesting things we can do," Il-Taeg Cho said.
The video below from ThermoFisher has strategies for isolating various parts of the cell and analyzing those samples with mass spectrometry. The video above is an explanation of how scientists fraction cells into different parts.
Sources: AAAS/Eurekalert! Via American Society for Biochemistry and Molecular Biology, Journal of Biological Chemistry