A Light Switch for Turning Proteins On & Off
A new tool invented by researchers at UC San Francisco enables the precise manipulation of the activity of proteins in cells. The technique uses a blue light to make proteins split apart, but in a way that is reversible. This work could have a tremendous number of applications; scientists study many different cellular processes and diseases by altering protein levels. There has been considerable interest that has led to myriad ways to tinker with cellular proteins, but there are hurdles to such work.
“A challenge of the post-genomic era is to figure out what specific proteins do in cells, and when and where they do it, something that cannot be achieved by genetic knockouts alone," explained Torsten Wittmann, PhD, a professor in the Department of Cell and Tissue Biology in UCSF’s School of Dentistry. “Here we show a new method to inactivate specific proteins acutely, locally and reversibly inside living cells, which should be able to address many of these questions."
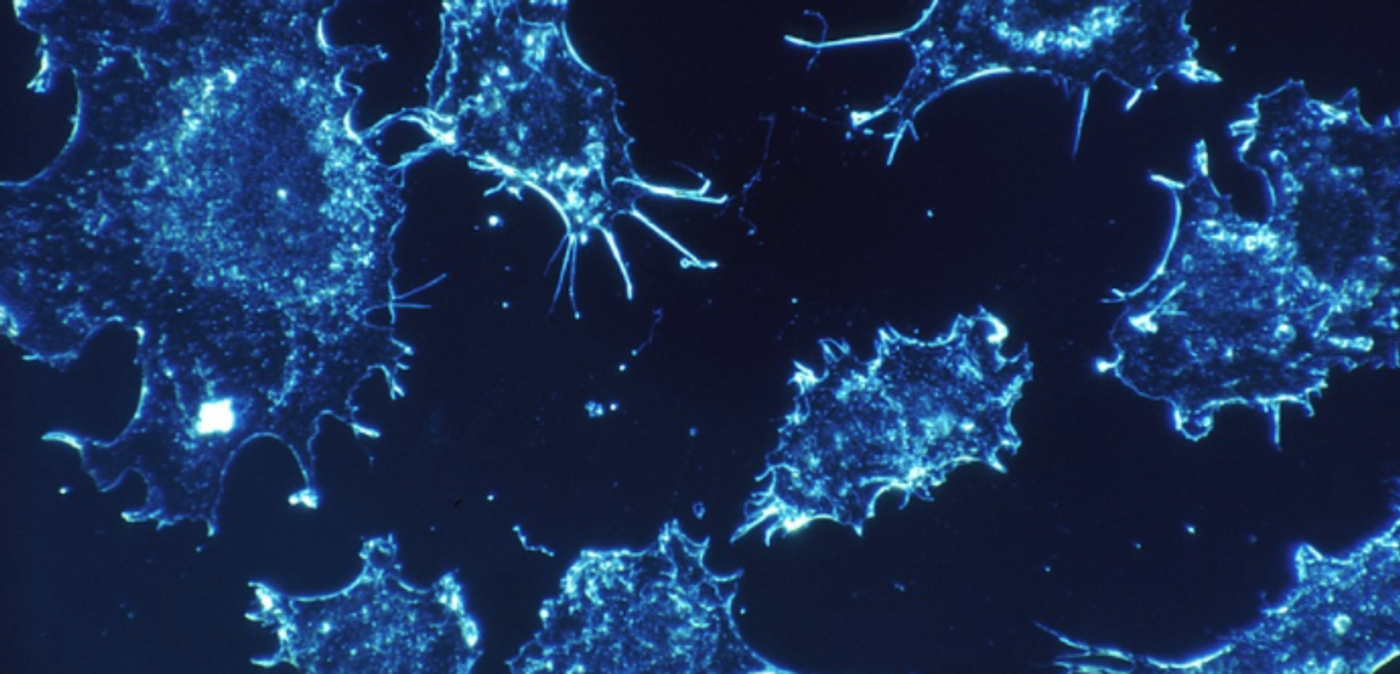
Reporting in Nature Cell Biology, the scientists used their invention to show that the movements of cancer cells are dependent on the architecture of the cell, its microtubules. They enable cells to shift in the direction of cell migration. This work could not only enrich our understanding of cell movement but also aid in the creation of therapies for cancer.
Optogenetics is a burgeoning field that uses light to control live cells. The photo-inactivation used for this study can make proteins light-sensitive in any cell; the function of proteins can be quickly turned off and on in specific parts of the cell.
The researchers took advantage of two proteins called LOV2 and Zdk1, they bind to each other in the dark but detach when they’re exposed to blue light. The investigators used genetic engineering to introduce the LOV2 and Zdk1 proteins into a larger protein, which then made that larger protein come apart when exposed to blue light.
Postdoctoral researcher and first author Jeffrey van Haren, Ph.D., used the tool to assess how microtubules influence the ability of a cell to move. LOV2 and Zdk1 were inserted into the EB1 protein, which regulates the growth of microtubules. When a blue light was shone on genetically manipulated cells, microtubule growth was decreased, and when the light was off, it returned to normal.
It had been thought that the actin cytoskeleton was the primary player in cell movement, said Whitman. This work indicates that the direction of a cell can be altered by halting the growth of microtubules toward the leading edge of the cell. Impressively, the investigators also trapped a cancer cell in a cage of light.
“We believe this strategy will be useful to be able to generate many other light-inactivated proteins,” Wittmann said. “A lot of cellular proteins are modular, with large folded domains tethered together by unstructured linkers. By inserting a photo-inactivation element in place of these linkers, we should be able to make any such protein sensitive to light."
This work could dramatically reduce the amount of time it takes to lower and raise protein levels. “Photo-inactivation lets us turn proteins off and back on in living cells in real time, and do so with much more spatial accuracy than has been possible before,” Wittmann noted. "I hope this becomes a key tool for anyone interested in understanding how these tiny molecular machines make every cell in our bodies tick."
Sources: UCSF, Nature Cell Biology