Smart Material that Can Switch Back-and-Forth Between Different Solid States
The research of metamaterials, the artificial entities that possess the properties unknown to nature, is at the forefront of engineering innovation.
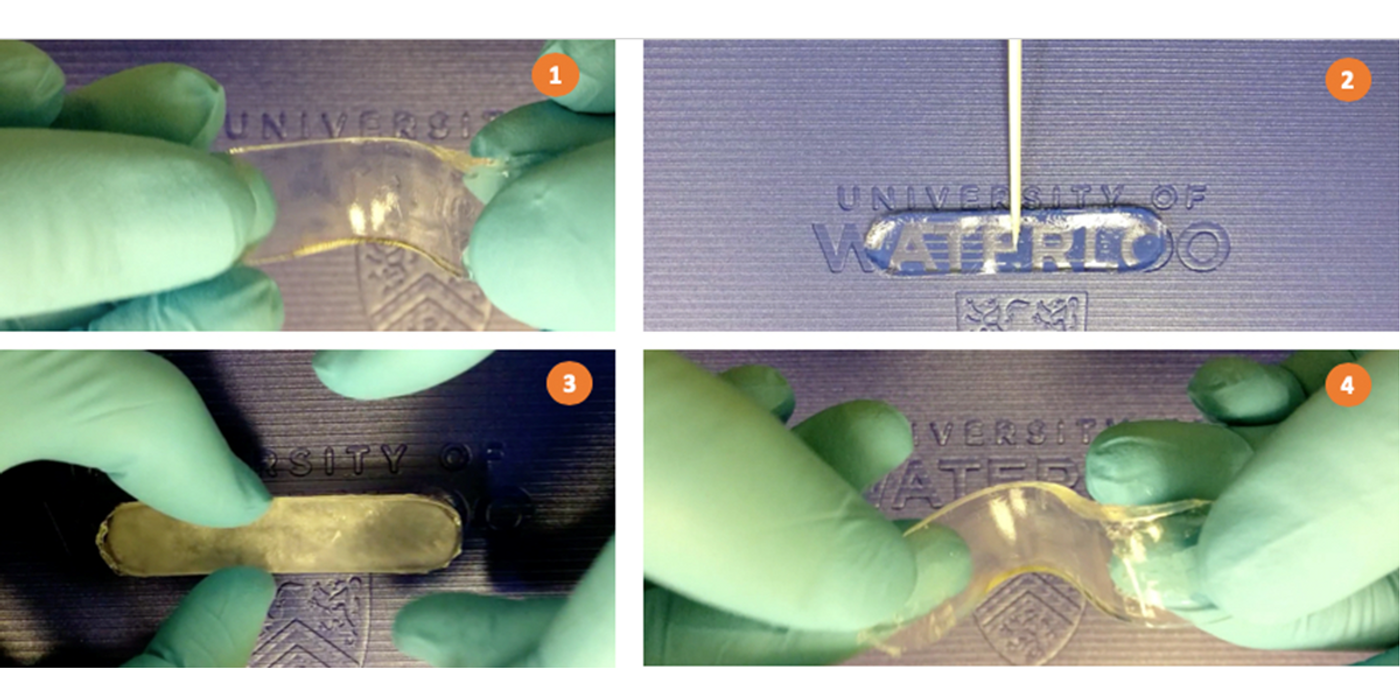
According to a recent publication, a team of chemical engineers at the University of Waterloo developed a new material can switch back and forth between two solid states: an opaque, hard solid and a soft gel. This breakthrough could spell revolutionary changes in soft robotics and adhesive applications.
Scientists have been pursuing materials with novel properties for decades. Some of the advanced materials consist of multiple composite elements (such as ceramics, metal matrix, or polymers) that are arranged in repeating patterns. These unique internal architecture enable them to behave like nothing else the world has seen. For example, the negative-index metamaterials are capable of bending light very differently from ordinary materials because of its negative refractive index. A collaboration project between MIT and Lawrence Livermore National Laboratory (LLNL) created a super-stiff, ultra-lightweight aerogel that can tolerate a load 160,000 times of its own weight.
The team of Canadian researchers, led by Dr. Boxin Zhao a professor of Chemical Engineering at the University of Waterloo, produced their state-shifting material by incorporating supercooled liquid salt into a polymer, forming a so-called sal-gel.
LabRoots reached out to Kuo Yang, a Ph.D. candidate and the first author of the publication through an email interview. He explained what inspired him to invent the hybrid material.
"Most research on gels have been focused on making the gel's polymer network more functional (i.e. 'smarter'). In contrast, I have always wondered: why not make the liquid in gels 'smarter'? With that in mind, the project was kickstarted one day when I thought it might be interesting to turn the sodium acetate (that I saw from heating pads) into a gel," he commented.
The soft-to-hard shift of the material requires a short, poke to the gel, a stimulation known as nucleation. Nucleation is the first step for the self-organizing crystals to start its phase transition. For instance, when sugar molecules become supersaturated in water, nucleation allows them to form large crystals through binding to one another. In its hard state, the material gets 104 times stiffer than its soft state. The return back to soft state can be realized through brief and gentle heating.
Kuo acknowledged that there are other functional materials capable of adjusting their stiffness by orders of magnitude: "The potential applications for our hybrid material generally overlap with existing shape memory or stiffness-changing materials."
"However, what makes our materials better than the existing ones is that our materials do not require additional energy to maintain stiffness states and therefore are more energy efficient," he added.
For their next step, the team of material engineers is hoping to address one of the shortcomings of this work--the supercooled liquid in the gel isn't as stable as they would like. "without lubricating the gel's surface, direct contact of the gel in its soft state can trigger unintended crystallizations. Consequently, one of the next steps is to identify better ways to stabilize the liquid," said Kuo.
This latest research is reported in the journal Nature Materials.
Want to learn more about metamaterials? Check out this video from Seeker.
These Metamaterials Go Beyond the Properties of Nature (Seeker)
Source: UWaterloo
[This article contains LabRoots original content]