Everyone loves to play with bubbles. They are fascinating and beautiful to watch with their swirling rainbow of colors and changing shapes. But what exactly is a bubble and why are its colors so dynamic?
A bubble is a thin film of soapy water surrounding a pocket of air. The thin film has three layers consisting of a thin layer of water in between two layers of soap molecules. The soap molecules are oriented so that the hydrophilic end faces the water layer, while the hydrophobic tail pushes away from the water. Bubbles might start out in different shapes but they all try to become a sphere. The spherical shape minimizes the surface area of the bubble, requiring the least energy to attain. And what about all those moving colors?
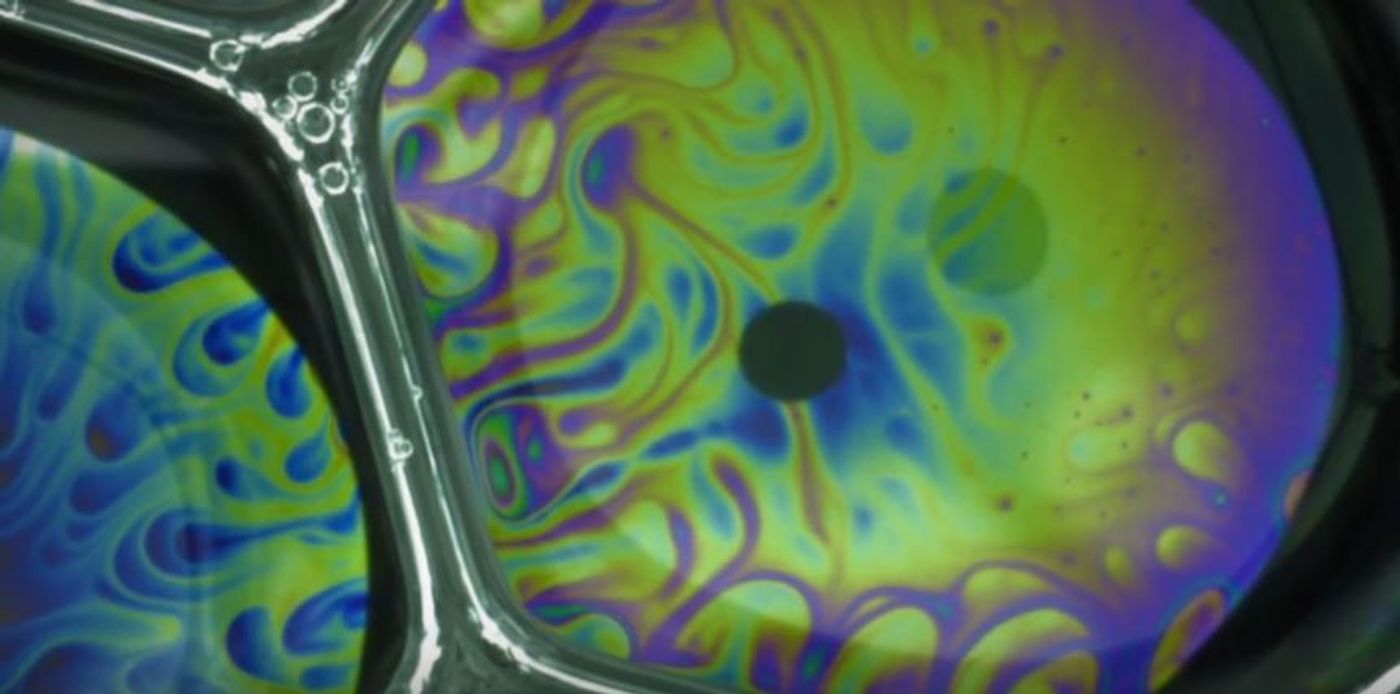
It turns out that this color movement has a purpose. Called the Marangoni effect, the whirling surface of bubbles is due to molecules moving from areas of low surface tension to higher surface tension. The effect helps to stabilize the bubble by distributing the surface tension evenly across the bubble surface.
Interestingly, bubbles have not always just been for play. Scientists across many different areas of research have looked into bubble dynamics and optics, from fluidics to engineering and biology. Joseph Plateau first studied the geometry of bubble surfaces in 1873, and Isaac Newton used bubbles to study optics. Most recently, bubbles have become an ideal model system to study fluid dynamics and liquid film technology.
Researchers in Chemical Engineering at Stanford University are studying the Marangoni effect in bubbles, and how it relates to liquid interfaces and surfactant systems such as human tears and lung tissue. The team, co-authored by Gerald Fuller, a professor of chemical engineering, has been able to pause the swirling in bubbles. Stopping this constant movement and balance in bubbles has never been accomplished before.
The study, published this month in
Physics Review Fluidics, explains the process of how the team was able to stop the flow of color on a bubble surface. First, a 1 mm air bubble is created in a soap solution. Then the bubble is raised to the surface quickly, then halted, and raised again. Every time the bubble is stopped on its way out of the solution a new layer of Marangoni flow is added to the surface, trapping the previous layer and halting its flow. By fluctuating the bubble movement, up to 7 layers could be created on a single bubble.
Each halted flow creates a new kaleidoscopic image of where each color is frozen in place. The frozen colors represent peaks and valleys of the surface of the bubble at that stage, allowing researchers to look closely at the surface tension balance.
“You almost expect that things that are mundane – day-to-day objects, simple things – are all figured out. You take it for granted,” lead author Saad Bhamla said. “If you ask the question ‘Why?’ even the simplest things have a lot that we can discover about them because we have better tools today, we have better techniques today.”
An understanding of this precise thin layer surface tension could play a role in future applications of stable surfactant foams in food products and engineered foams for medical treatments. The Fuller lab already studies one such treatment, a replacement for lung surfactants used to prevent collapsed lungs. The stabilization or destabilization of bubbles might seem simple but in practice can affect tools and techniques across many disciplines.
Sources:
Stanford News,
APS,
APS Video