Peptide Drugs Could Live a Longer (Half-) Life with This Chimera Ligand
Peptide and protein drugs like insulin are critical medications. Most of them cannot be taken orally because they will be digested in the intestine and broken down into amino acids and simple derivative compounds. When injected intravenously, peptides are subjected to fast excretion by the kidney and suffer from enzymatic degradation in the blood circulation, which results in short plasma half-life and a limited therapeutic time frame.
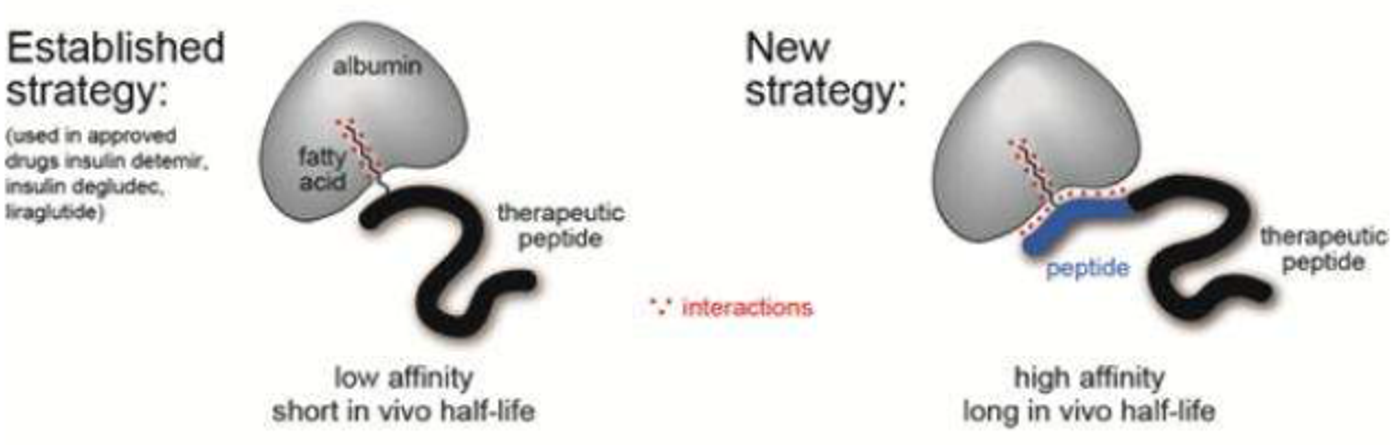
Modifications of peptides and proteins have proven to prolong their longevity in the blood. Shortening the amino acid chain and replacing L-analogue amino acids with D-amino acids can help to evade attack and cleavage by proteases in the blood. Alternatively, adding a fatty acid or short peptide moiety to the native polypeptide chain can allow the conjugated molecule to hitch a ride with serum proteins like albumin and stay in circulation longer.
Fatty acid ligands can extend the circulation half-life by helping the peptide piggy-back onto albumin. Take insulin detemir for example; it is a long-circulating human insulin analogue tagged with a myristic acid (a saturated fatty acid with a 14-carbon chain). It lasts for 5-7 hours in circulation. In comparison the tag-free insulin has only a 4-6 minute half-life. Most fatty acid-conjugated peptides show weak to moderate affinity for albumin. On the other hand, peptide-based ligands showed better affinity to albumin. However, they render the modified molecule hard to dissolve and distribute in blood, which significantly undermines the therapeutic effects of peptides.
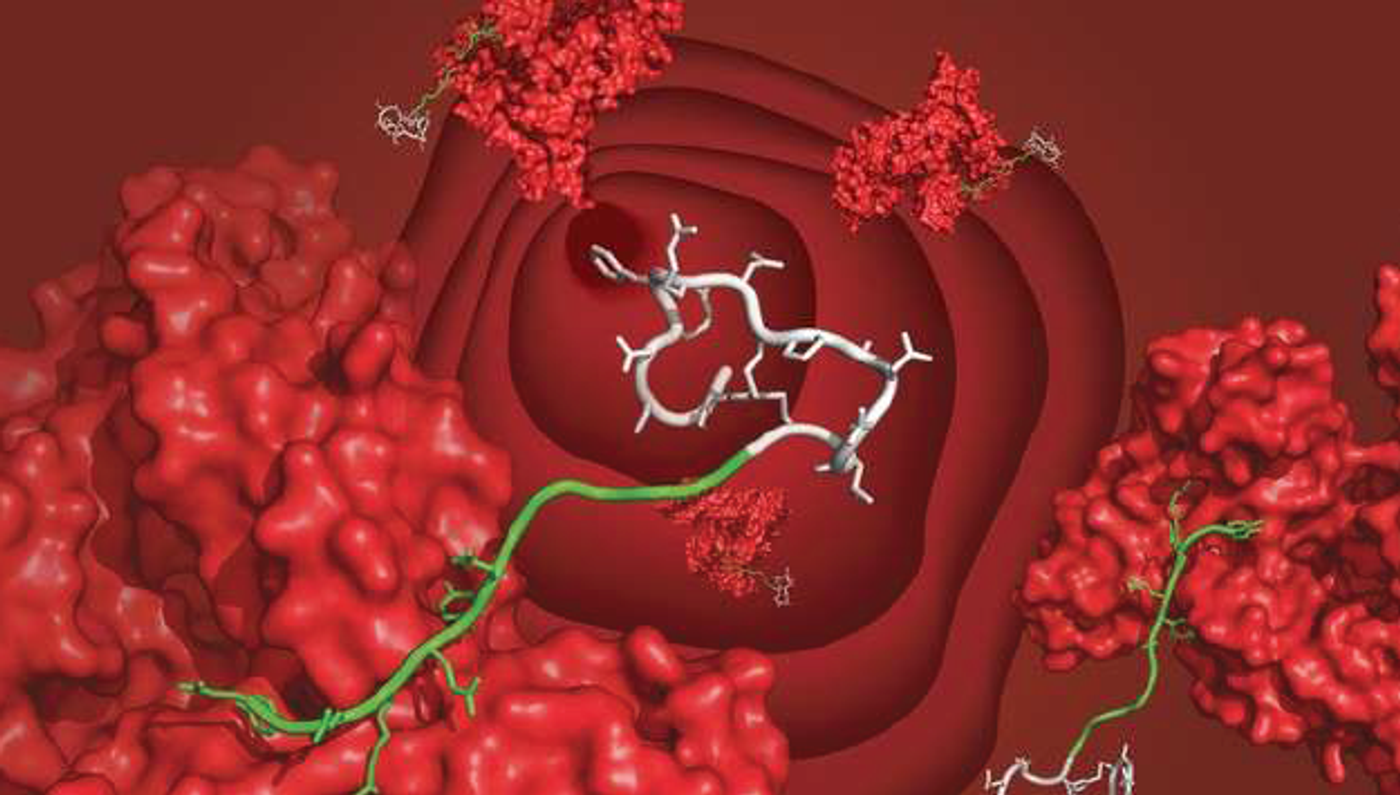
The chimera ligand (green), which is conjugated with a model drug peptide (white), is seen bound to serum albumin (red) in the schematic drawing. Credit: C.Heinis/EPFL
To solve this conundrum, a team of Swiss researchers led by Dr. Christian Heinis at the École Polytechnique fédérale de Lausanne (EPFL) has now developed a chimera ligand, a fatty acid fused with a second short peptide, which combines the advantages of two moieties. In their design, the fatty acid alone can bind to albumin with a moderate affinity and with a low micromolar Kd (dissociation constant – smaller Kd means higher affinity), and the peptide moiety provides extra interaction with albumin to bring the Kd down to the nanomolar level. Through elaborate synthesis and screening of various candidates, they found one that binds to human albumin with high affinity (Kd=39 nM), is highly soluble, and can easily be tagged onto peptides via standard synthesis procedure. To verify the functionality of their design, the researchers tagged two bioactive peptides, PK128 and UK18, both of which are protease inhibitors that suffer from fast renal clearance. The half-life of the two test peptides was significantly prolonged over 25-fold, though one of them suffered a drop in its inhibitory activity.
The comparison of ligand designs. Credit: Ecole Polytechnique Federale de Lausanne
When speaking about the potential of the new ligand, the group leader expressed his hope for its application on a bigger scale. "We expect that the tag presented in our work will interest a larger research and business audience because it is applicable to virtually any peptide moiety, including small proteins. The ligand can be appended to any peptide during solid-phase peptide synthesis on standard synthesizers, making it easily accessible for academic and industry labs," said Dr. Heinis.
Source: Phys.org/Nature Communication