A "Golden" Key to Unlock the Door to Novel Medicinal Chemistry
A joint team of chemists from UC Berkeley and Lawrence Berkeley National Laboratory came up with a novel method for adding trifluoromethyl group to molecules, according to a recent publication in the journal Science.
The trifluoromethyl group, or CF3-, is a highly popular substituent in synthesizing bioactive compounds because the covalent bond between carbon and fluorine is the second strongest single bond. It has been proven to be resistant to metabolism, therefore improving the circulation time for drugs.
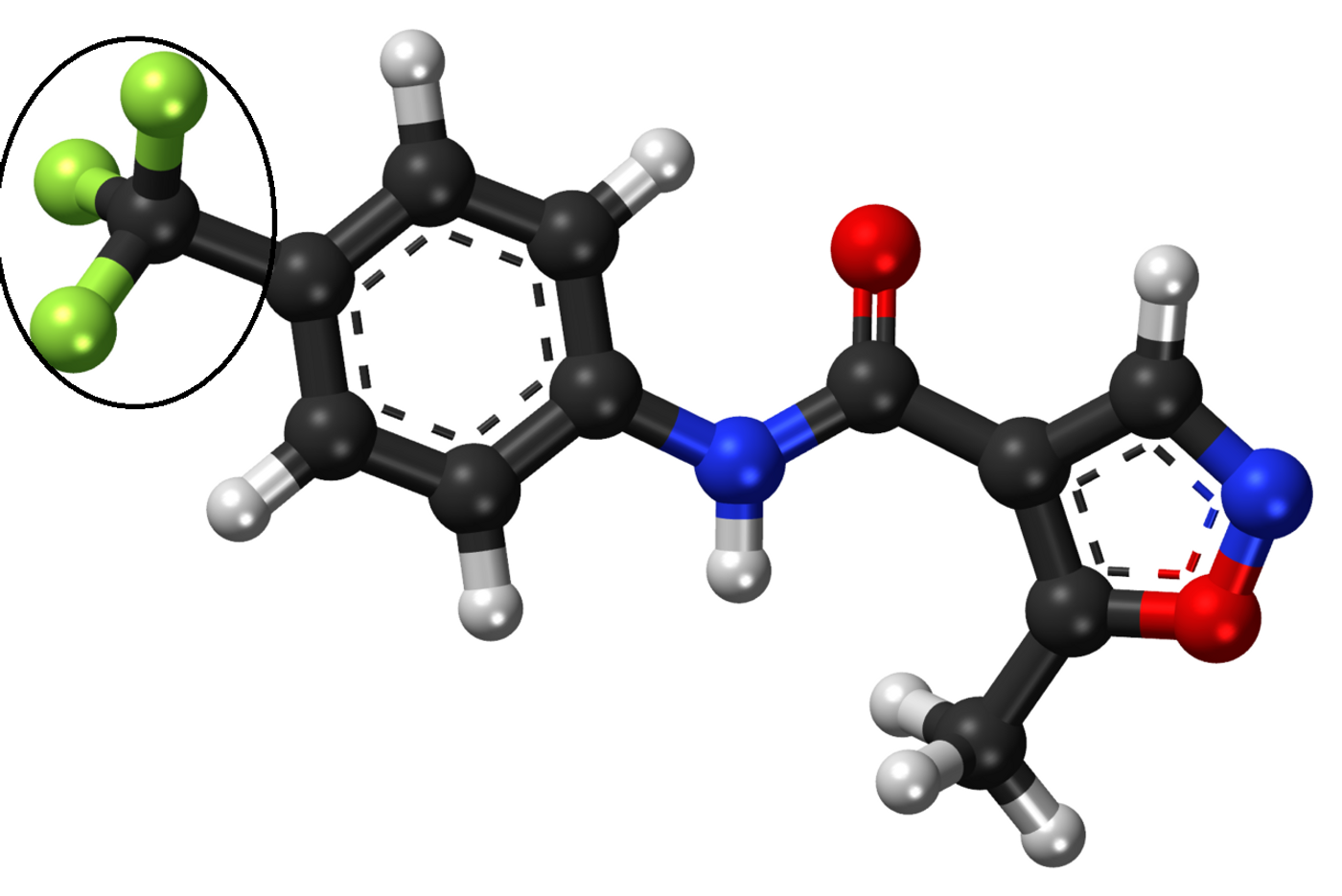
Leflunomide molecular structure displayed in a ball-and-stick model; trifluoromethyl group in circle. Credit: Wikipedia
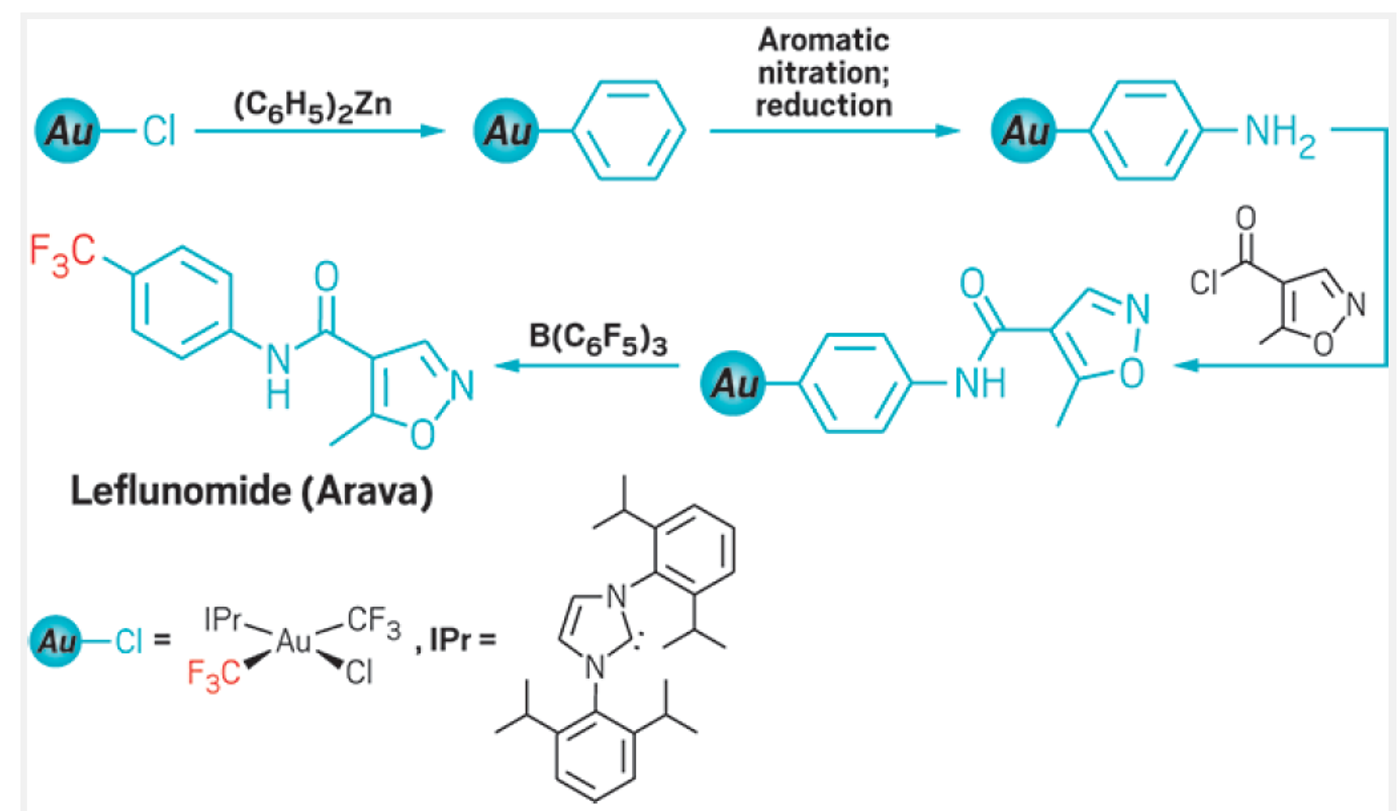
While working on something totally different, the researchers stumbled upon a trifluoromethylation compound as the reaction product. By looking carefully into the process, they discovered a so-called fluoride rebound mechanism: in the reaction, a borane catalyst grabs a fluoride from the CF3- group in a gold complex. The activated CF2 fragment is then free to bond with various types of carbon substituents, which share the gold atom as the center. Once a fluoride returns to the CF2 fragment, the trifluoromethylated compound parts ways with the metal complex. Because the bond between gold and carbon tolerates different reaction conditions including some very harsh ones, it makes this method extra valuable for the synthesis of a variety of medicinal molecules. To explore their reaction's potential the researchers synthesized Leflunomide a rheumatoid arthritis drug using the novel mechanism. They also took it a step further and adapted the reaction to generate radioactive tracers, so that fluorine-18-based radiolabeled diagnostic probes (for positron emission tomography <PET>) can be synthesized.
The fluoride rebound mechnism in the synthesis of leflunomide a rheumatoid arthritis drug. Credit: C&EN
The authors did not think the original idea would manifest in this direction. “None of us started this project with PET in mind. But now, because we stopped to investigate an unexpected result, we have a platform from which we can start to develop new tracers, not to mention a new mechanism to help us think about synthetic problems,” said Mark D Levin, the first author.
What are catalysts? Credit: FuseSchool