Strong Like Aluminum
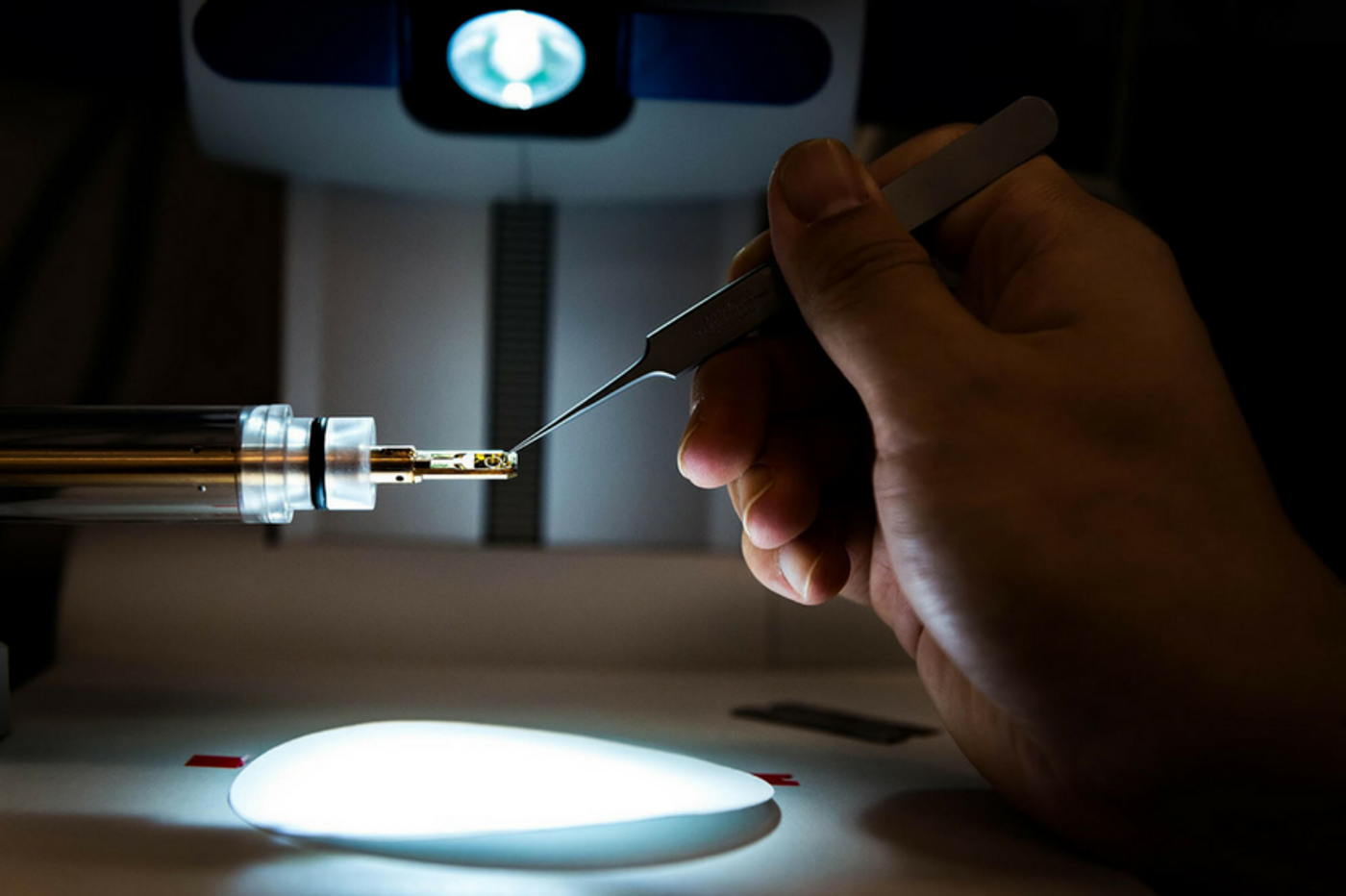
Material engineers from Purdue University announced that they have generated a super-strong aluminum-iron alloy. According to their recent publication in the journal Advanced Materials, the alloy is so strong that it can rival "the strength of stainless steel".
Light and strong metal alloys are the most sought-after crown jewel in the industries such as the automobile, electronics, military, and aerospace. For example, one of the major trends in the automobile industry is the lightening of vehicles. Using materials like carbon fiber, lighter metals and metal alloys that are stronger and lighter than steel, auto manufacturers have built a new generation of vehicles that are substantially lighter and environmentally friendlier than previous ones.
Aluminum alloys are often known for their lightweight but not strength. The Purdue engineers introduced a special structural feature called “9R phase stacking faults” into their alloy enhance its strength. Try picturing this: in the nanoscale, metal atoms are stacked together in a repeating sequence to form a scaffold-like structure, officially known as a crystal lattice. A stacking fault is a type of defect which characterizes the disordering within the planes of the scaffold. The 9R phase stacking fault is of particular interest to the Purdue engineers: it consists of a repeating unit of nine atomic layers—six fault planes plus three normal planes.
While stacking faults have been achieved and known improve the strength and ductility of other metals like copper and silver, no one has yet managed to do the same in aluminum. Forming stacking faults within aluminum lattice has proven especially difficult due to its high stacking fault energy. The material researchers introduced the 9R phase in aluminum by introducing iron atoms into aluminum's crystal structure via a procedure called magnetron sputtering.
The resulted aluminum-iron alloy have an extra feature: nanotwinning, which have been the focus of intense research lately. Twin boundaries occur when two metal crystals of the same type intergrow in the nanoscale, which leads to a minimized misorientation between them. Nanotwinned metal crystals have enhanced mechanical strength while maintaining good ductility, electrical conductivity, and thermal stability in metallic materials.
Commenting on one of the strongest aluminum alloys ever created, Xinghang Zhang, a Purdue Engineering professor and the study's senior author said: “Molecular-dynamics simulations, performed by professor Jian Wang’s group at the University of Nebraska, Lincoln, showed the 9R phase and nanograins result in high strength and work-hardening ability and revealed the formation mechanisms of the 9R phase in aluminum,” Zhang said. “Understand new deformation mechanisms will help us design new high strength, ductile metallic materials, such as aluminum alloys.”
Super-strong Aluminum Alloy. Credit: Purdue Engineering
Source: Purdue University