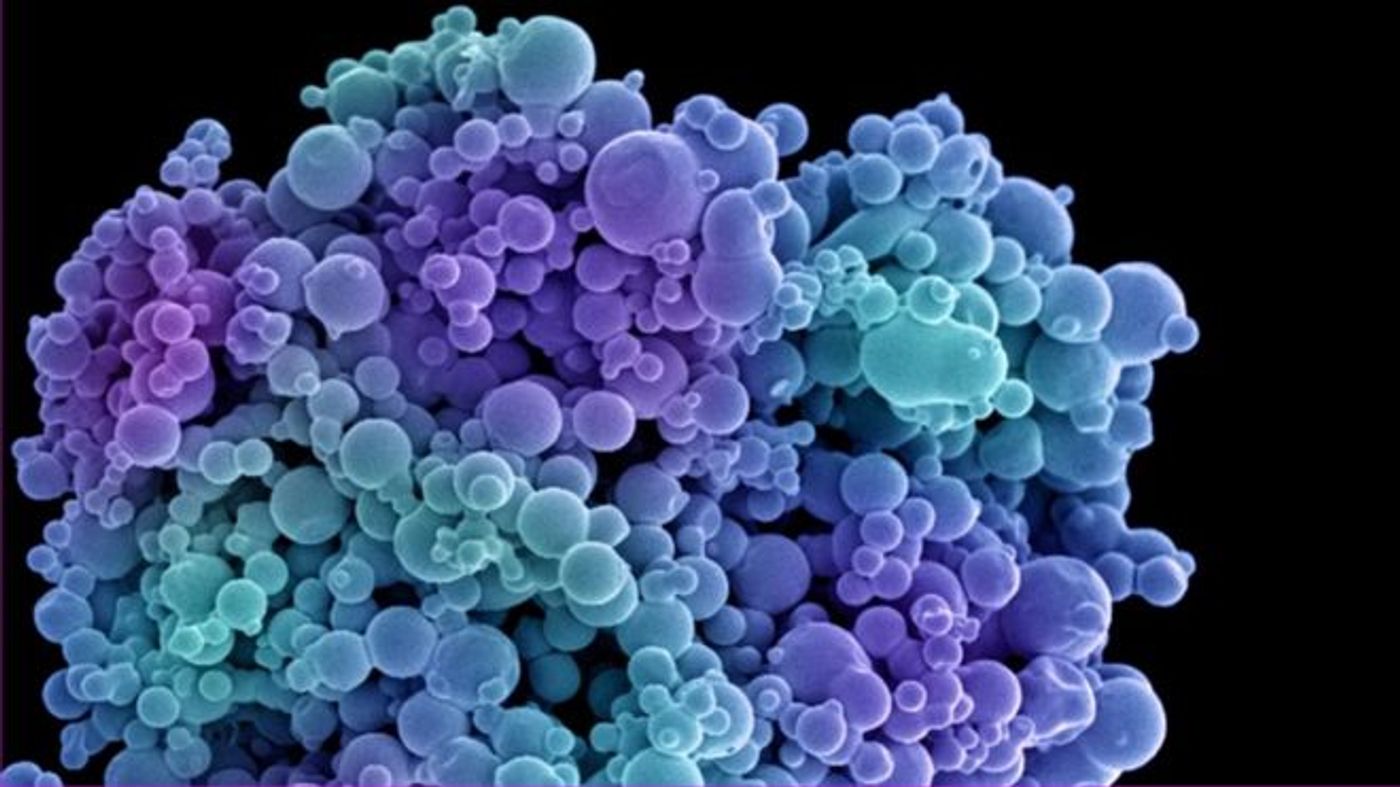
In the battle against cancer, scientists may be one step closer to detonating the tumor base camp, literally.
The research, presented at the UK’s National Cancer Research Institute Conference, describe the
engineering of nanoparticle “grenades” set to
blast tumors upon heat activation.
In the current treatments of cancer, removal is still a first line of defense, followed by bursts of high-dose radiation and chemotherapy. While these treatments have dramatically changed the outcomes of many cancer prognoses, they are invasive and often cause collateral damage to the patient’s healthy cells. As such, the new goal has been to not only create highly effective anti-cancer drugs, but also to deliver it in such a way that targets only the cancer cells for destruction.
Scientists quickly realized the goal for high efficacy in cancer treatment can be achieved with nanotechnology, utilizing microscopic lipsomes to deliver the potent drugs directly to the cancer cells.
"Liposomes are small bubbles of cell membrane that act like a cellular postal service, delivering molecules to our cells. Using them to deliver cancer medicines has been a holy grail of nanomedicine. But finding ways to accurately direct the liposomes towards tumours has been a major challenge in targeted drug delivery.” - Professor Charles Swanton, chair of the 2015 NCRI Cancer Conference
The current research adds the much desired layer of specificity of the drugs, using temperature-sensitive liposomes (TSL). The TSL are uniquely modified to be leaky and explode upon landing on tumor hotspots. At 37C, the normal body temperature, the water-tight TSLs are chemically and thermally stable. However, at 42C, the “pin is effectively pulled and the drugs are released,” says Kostas Kostarelos, study author and Professor of Nanomedicine at the University of Manchester. Upon exposure to 1 hour of mild heating at 42C the team reported significant enhancement of cytotoxic activity in human melanoma cancer cells. The effects of the TSLs also carry to animal studies, showing improved survival in mice carrying tumor transplants.
If five degrees of separation is apparently sufficient to discriminate between healthy and cancer cells, then how do we raise the temperature of only the tumors and not the whole body? For tumors near the body surface, such as head, neck, and skin, a simple heating pad would do the trick. Internally located tumors would require use of a heat probe or ultrasound, which are already being used in the clinic.
The benefits of a heat-triggered TSL grenade could be multifold. First, because of the specific temperature requirement for activating the TSL, the anti-cancer drugs should only target heated cancer cells. This could mean increased efficacy of the anti-cancer drugs and more effective annihilation of the tumors. Secondly, because normal cells are not affected, this could mean significantly less collateral damages, reducing unwanted side effects for patients.
The potentials could be huge: TSLs could offer a way to achieve faster, safer, and longer-lasting remissions from cancer.
"These studies demonstrate for the first time how they can be built to include a temperature control, which could open up a range of new treatment avenues. This is still early work but these liposomes could be an effective way of targeting treatment towards cancer cells while leaving healthy cells unharmed." - Professor Charles Swanton
Watch the video to learn more about how nanoscale liposomes work!
Sources:
BBC,
NCRI