The Fight Against Brain Cancer Shrinks to 20 Nanometers
What happens when a nanoscience polymer expert, a biomedical engineer, and a couple of surgeons get together? They come up with a new potent drug delivery system for one of the most intractable brain cancers: glioblastoma multiforme.

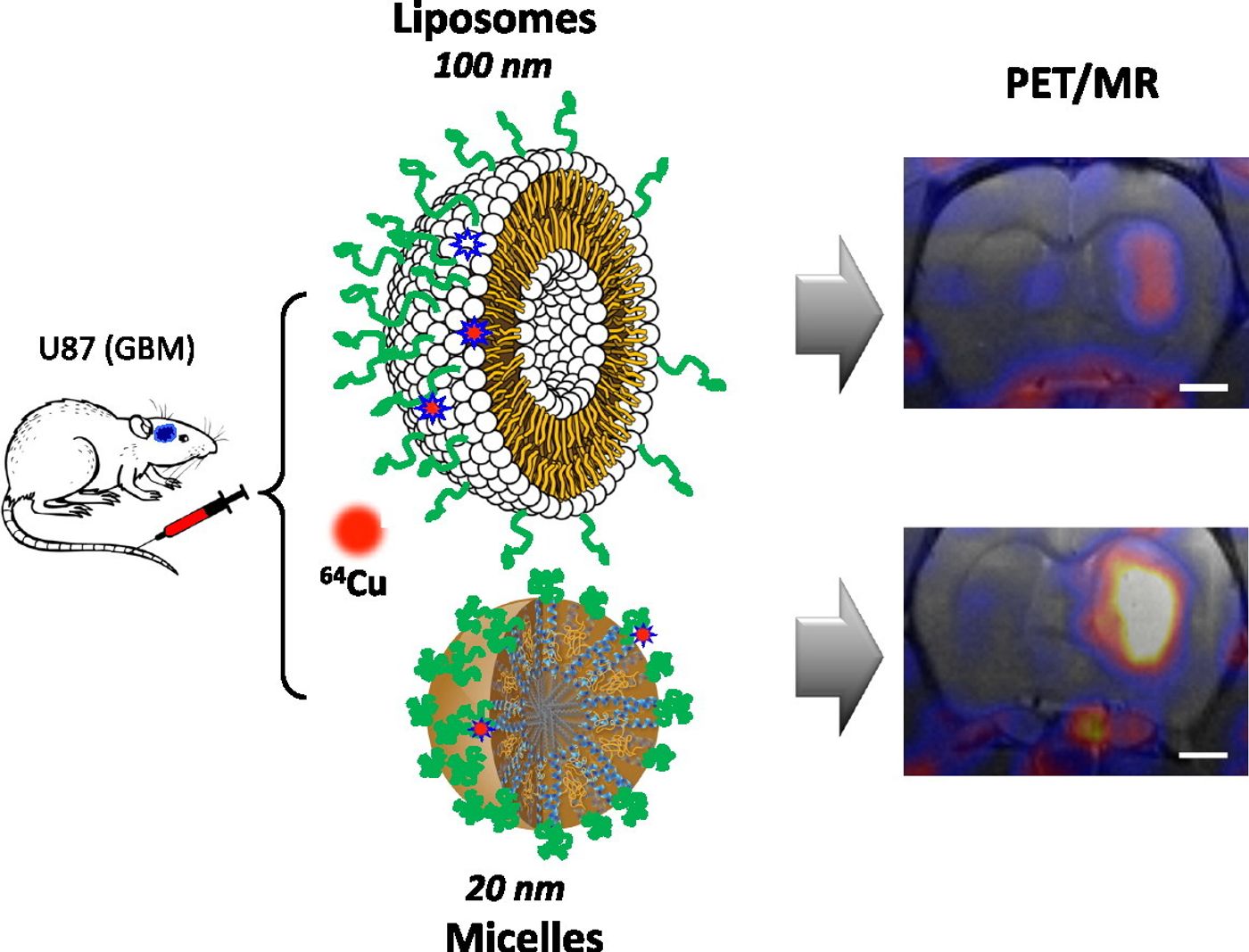
The new drug delivery system comes from the University of California, Berkeley, where researchers have developed a new family of nanocarriers that’s able to breach the blood brain barrier. The carrier, nicknamed “3HM,” is a 3-helix micelle sphere that’s a mere 20 nanometers in diameter. To put it in perspective, the 3HM particle at is nearly 4000 times smaller than the diameter of a human strand of hair, which is 75,000 nanometers. The 3HM is produced from the self-assembly of proteins and polymers that are amphiphillic. This means that in addition to being extraordinarily tiny, the 3HM particle is both water- and fat-loving, features that imbue an unusually stable kinetic profile to the nanoparticle.
Once they developed 3HM, they soon realized that these combined features make the 3HM highly unique and well suited for crossing the blood brain barrier. “We looked into critical needs for nanocarriers with these attributes and identified the treatment of glioblastoma multiform cancer as a potential application,” said Dr. Ting Xu, lead nanoscientist.
To test if 3HM can cross the blood-brain barrier and where the particle is distributed, the team did a trial in rats with grafted glioblastoma multiform (GBM) tumors. They also tested the performance of 3HM against a current FDA-approved nanocarrier that’s more than 5 times larger than 3HM. Both types of nanocarriers were labeled with a radioactive form of copper and injected into the GBM rats. The team traced the localization of the copper-labeled nanocarriers with positron emission tomography (PET) and magnetic resonance imaging (MRI). Indeed, the 20-nanometer 3HM carrier not only passed through the blood-brain barrier effortlessly, but it also collected evenly at the tumor sites at nearly twice the concentration of the larger 110-nanometer nanocarriers. Even more impressive, the team reported that the nanospheres did not clear readily from the tumor, showing the infiltration is deep, specific, and long lasting.
It is not hard, then, to imagine the outcome for GBM treatment if 3HM actually ferried a cargo of cancer drugs in its spheres. So far current surgery, post-surgical chemotherapy, and radiotherapy have all proven vastly ineffective for treating GBM. It’s nearly impossible to surgically remove all the cancerous tissues in the brain because the malignant glial cells form vast networks of tendrils throughout the brain, earning GBM the name “octopus tumors.” Treating GBM with anti-cancer drugs has also been nearly futile because the drugs loose its efficacy due to the blood brain barrier. Even with standard care, the median survival for GBM diagnosis is just over 1 year; the five-year survival rate is less than 10%.
Researchers are hopeful that the 3HM nanocarrier will dramatically improve these odds for GBM patients. The 3HM nanocarrier could safely ferry anti-cancer drugs across the blood brain barrier without the drugs being inactivated. Further, because of its exceedingly small size, 3HM shows deep and localized infiltration inside the tumor. This could mean that higher potency can be achieved with smaller doses, killing the cancer while minimizing the side effects to the patients.
Additional Source: Berkeley Lab