The beloved actor Gene Wilder died earlier this week from complications of Alzheimer’s disease. He was one of the tens of thousands of people who succumb to the disease, according to the Centers for Disease Control and Prevention.
But there’s some hope peeking through the grim statistics of this disease. Scientists recently announced highly
promising results of a drug that purportedly can erase the toxic protein clumps in the Alzheimer brain.

The drug, known as aducanumab, targets the amyloid beta plaques in the brains of animal models, and now also in human clinical trials. As this drug progresses to Phase 3 to test efficacy in a larger group of patients, researchers are hopeful it will become the first drug to actually reverse Alzheimer pathology.
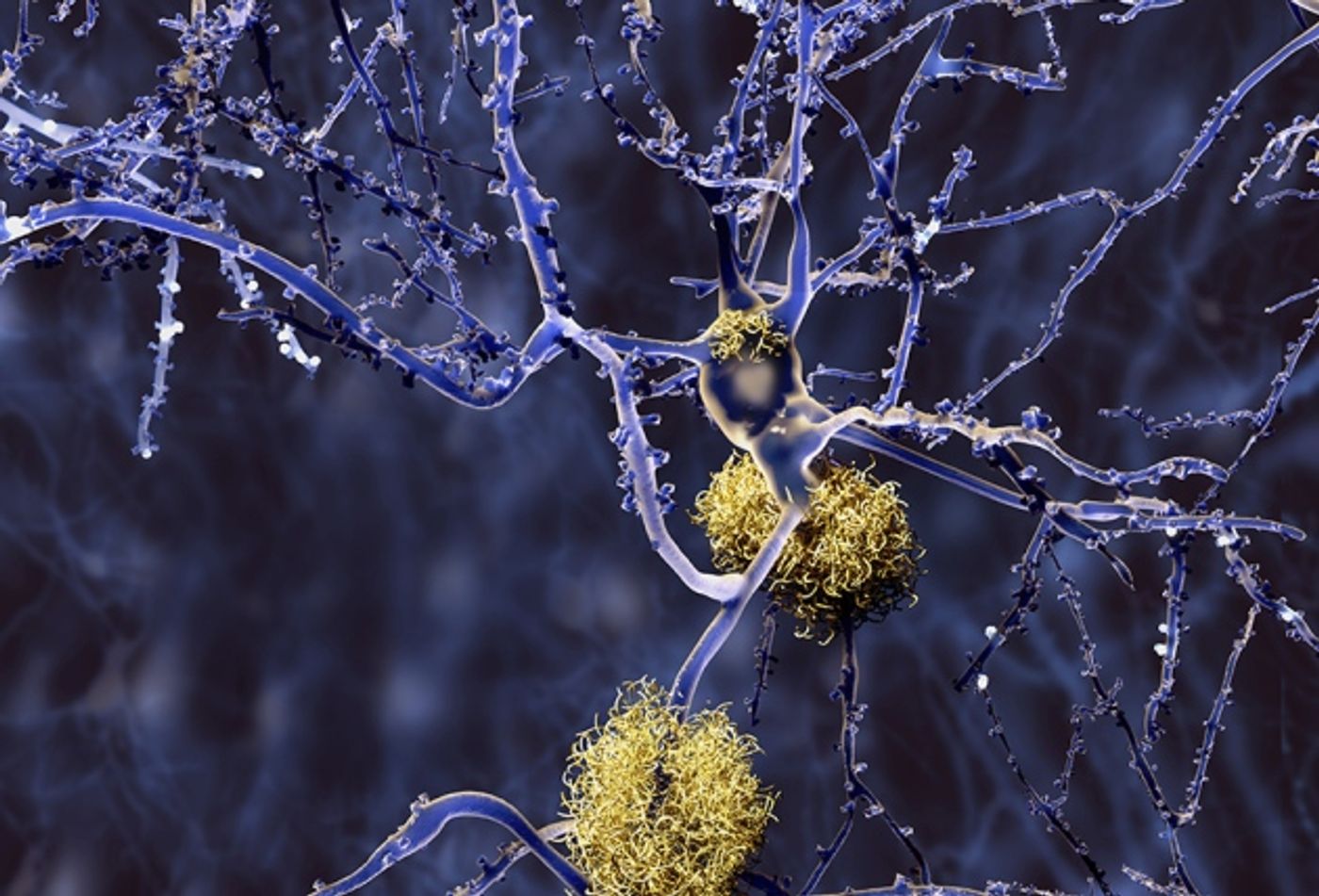
Alzheimer’s disease is pathologically defined with the presence of amyloid beta protein clumps in the brain. These are toxic to neurons and interfere with memory, cognition, and other normal functions of the brain. The CDC estimated 85,000 people died from Alzheimer’s disease in 2015; however, this number is believed to be an underestimate, as many deaths may not be correctly attributed to Alzheimer’s.
Aducanumab is an immune compound that appears to stimulate the body to remove the toxic amyloid clumps. In the latest study, scientists split 165 patients with mild Alzheimer’s disease into two groups: those who received aducanumab and those who received a placebo. This was done for 1 year, with the intent on demonstrating that the drug was safe to take.
Indeed, researchers found encouragement beyond safety. Through analyzing brain scans, the team found that patients treated aducanumab had less amyloid plaques than those treated with the placebo.
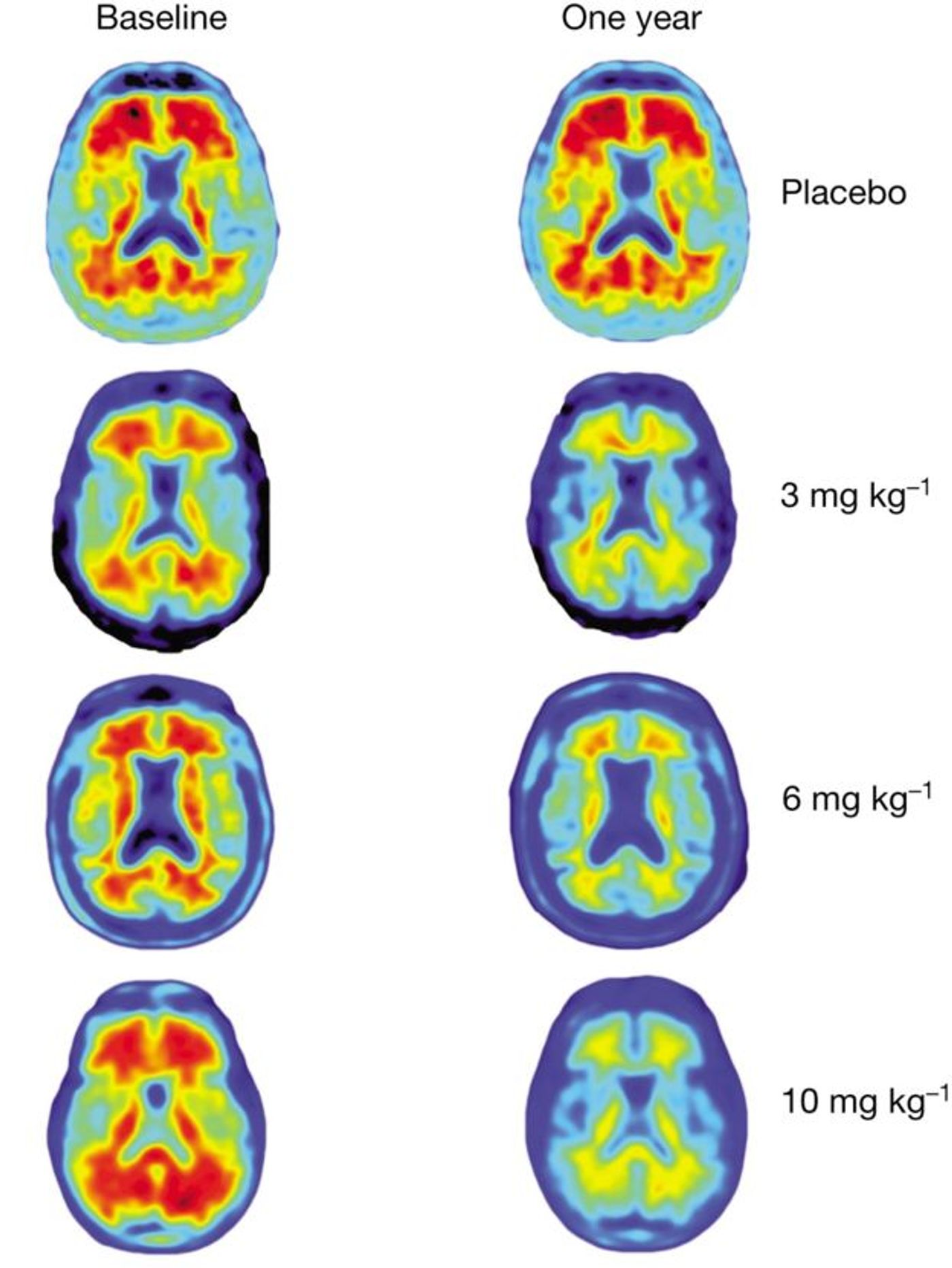
"After one year, you can see no red on the image, meaning the amyloid has almost completely
disappeared," said Dr. Roger Nitsch, a co-author of the study and the director of the Institute for Regenerative Medicine at the University of Zurich.
In addition, there was a direct correlation between dose and effect – the higher the dose, the greater the clearance of the toxic protein clumps. Notably, however, the higher dosages were also associated with more side effects.
Though the purpose of the study was to establish safety, the team did carry out some memory tests with some promising results. "We believe that's a hint of efficacy," study co-author Dr. Alfred Sandrock, a neurologist and an executive vice president at Biogen,
"We're encouraged that, there appeared to be a slowing of cognitive decline at a dose-dependent manner, and also a dose-dependent slowing in functional decline," said study co-author Dr. Stephen Salloway.
The next phase of the study will definitively answer whether aducanumab holds cognitive benefits for Alzheimer’s patients. A total of 2,700 patients are slated to be enrolled in this next phase, which will be an international effort involving North America, Europe, and Asia.
Additional sources:
BBC,
Nature News

 Alzheimer’s disease is pathologically defined with the presence of amyloid beta protein clumps in the brain. These are toxic to neurons and interfere with memory, cognition, and other normal functions of the brain. The CDC estimated 85,000 people died from Alzheimer’s disease in 2015; however, this number is believed to be an underestimate, as many deaths may not be correctly attributed to Alzheimer’s.
Alzheimer’s disease is pathologically defined with the presence of amyloid beta protein clumps in the brain. These are toxic to neurons and interfere with memory, cognition, and other normal functions of the brain. The CDC estimated 85,000 people died from Alzheimer’s disease in 2015; however, this number is believed to be an underestimate, as many deaths may not be correctly attributed to Alzheimer’s.