Post-traumatic stress disorder (PTSD) affects millions of people, but the disorder is notoriously difficult to treat. Even with our best treatment efforts, researchers say a significant portion of patients are still left behind.
But recently, the psychedelic MDMA, otherwise known colloquially as “ecstasy,” is gaining traction as an unconventional but effective treatment for PTSD. Furthermore, the FDA also saw the potential in this drug, and has approved it in a large-scale clinical trial for PTSD. The move could pave the way for legally prescribed “ecstasy” as a psychotherapy agent in the future.
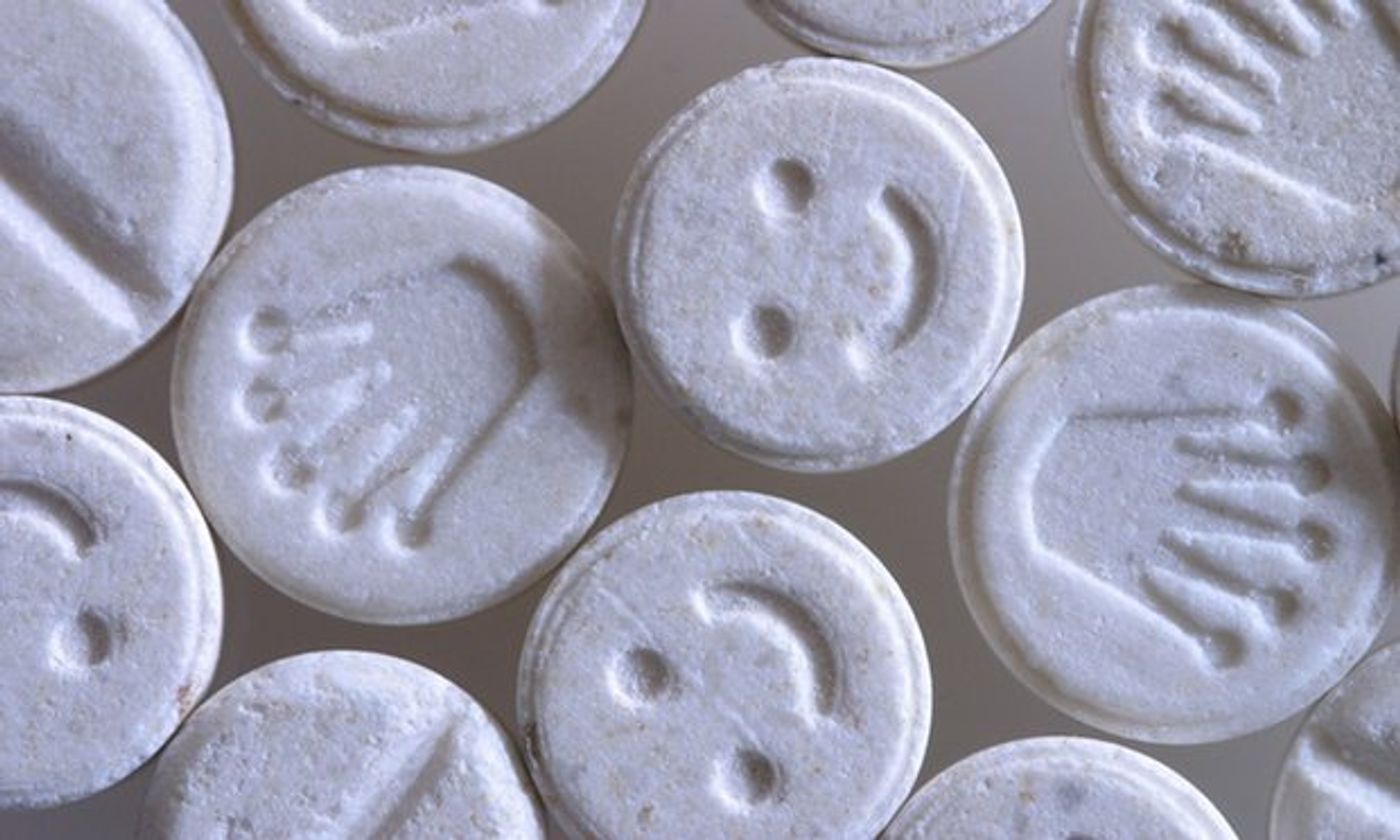
Known on the street as “E,” “Molly,” and “millennial aspirin,” MDMA (3,4-Methylenedioxymethamphetamine) is a popular recreational psychedelic drug. The compound acts on the amygdala to induce feelings of euphoria, empathy, and otherwise heightened emotions.
Heavy abuse of the drug – leading to addiction, memory issues, paranoia, and other adverse symptoms – led the FDA to classify it as a Schedule 1 substance and its eventual ban. However, many researchers saw potential in MDMA as a psychotherapy drug, precisely because of how it affects the brain.
In a few early stage clinical trial of MDMA, patients reported working through their trauma in ways that could not be replicated with conventional treatments. One study found a 56 percent decrease in the severity of their PTSD symptoms with the use of MDMA. This shift led more than two-thirds of the patients to free themselves from the diagnosis of PTSD – a remarkable outcome considering the ineffectiveness rate of current therapies. Furthermore, the results seem to have a lasting effect, even after the drug was discontinued.
“The body of evidence that shows MDMA works is so strong and so encouraging,” said Rick Doblin, the founder and executive director of Multidisciplinary Association for Psychedelic Studies (MAPS), which is sponsoring the current Phase III clinical trial, involving over 200 participants.
In addition to obtaining the unprecedented OK from the FDA to use MDMA in the clinical trial, the team also applied for the “breakthrough therapy” status. This designation is typically reserved for drugs that can “treat a serious or life threatening disease or condition.” If granted and trial results prove promising, MDMA can be on the fast track for approval as a PTSD psychotherapy drug by as early as 2021.
Of note, researchers underscore that the trial is to expand the psychedelic’s medicinal uses, not its recreational potential. So to people who doubt the safety of legal ecstasy, Doblin says: “This is never going to be a take-home drug. It’ll only be prescribed by doctors with special training, for people with specific conditions.”
Additional sources:
NY Times,
Popular Science
 Known on the street as “E,” “Molly,” and “millennial aspirin,” MDMA (3,4-Methylenedioxymethamphetamine) is a popular recreational psychedelic drug. The compound acts on the amygdala to induce feelings of euphoria, empathy, and otherwise heightened emotions.
Known on the street as “E,” “Molly,” and “millennial aspirin,” MDMA (3,4-Methylenedioxymethamphetamine) is a popular recreational psychedelic drug. The compound acts on the amygdala to induce feelings of euphoria, empathy, and otherwise heightened emotions.