Drug Reduced Recurrent Ventricular Tachycardia

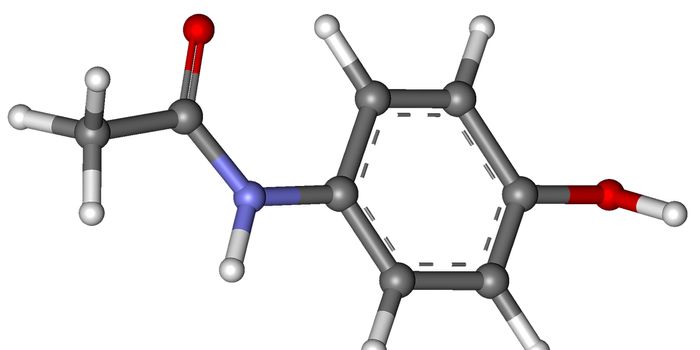
A common drug used to treat chest pain, known as ranolazine and branded as Ranexa®, was found to be safe in a clinical study of more than 1,000 patients with implantable cardioverter defibrillators (ICDs) and at high risk of ventricular tachycardia (VT) as well as ventricular fibrillation. The studied patients had an average age of 64 with women making up 18% of the participants. Although the drug did not effectively reduce the risk of the first occurrence of ventricular tachycardia, ventricular fibrillation, or death in a high-risk population—it has lowered the number of recurring episodes of ventricular tachycardia.
"This drug is already on the market to treat chest pain, and while it didn't provide the revolutionary results we had hoped for, I think there's a place for it in the toolbox of treatments for select patients," explains Wojciech Zareba, M.D., Ph.D., the lead author of the study and professor of Cardiology at the University of Rochester Medical Center. "Ventricular tachycardia is the most common arrhythmia that individuals with ICDs experience. This drug could be an option for patients who haven't had success with other therapies."
Results of the clinical study were recently published by researchers at the University of Rochester Medical Center in the Journal of the American College of Cardiology. The study, sponsored by the National Institutes of Health, was inspired by previous work strongly suggesting that ranolazine may reduce the incidence of arrhythmias. The clinical trial was also motivated by the need to seek more options for anti-arrhythmic therapies.
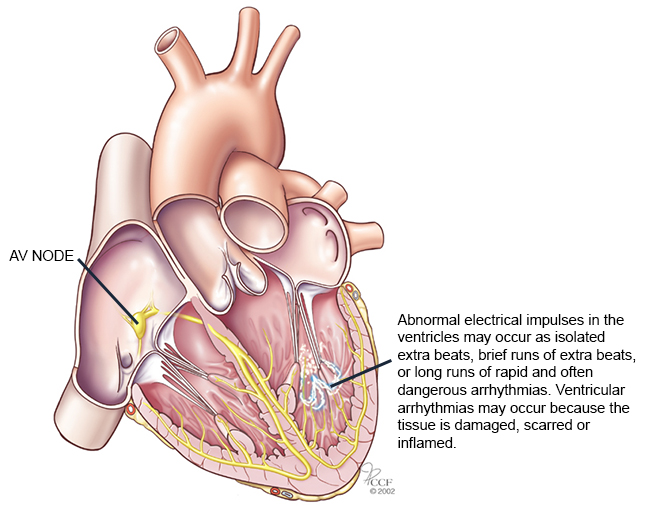
Ventricular tachycardia consists of a fast and abnormal heart rate originating in the ventricles (lower chambers of the heart) and can lead to dizziness, lightheadedness, palpitations, and loss of awareness. Results propose the potential role of ranolazine in patients with ICDs who cannot tolerate other treatments and aren't eligible for ablation; a procedure aiming to correct faulty heart rhythms. Further analysis has also shown that ranolazine reduced recurrent ventricular tachycardia by 30 percent than patients with the placebo. "Through this trial, we learned that ranalozine can be administered safely in patients with life-threatening abnormal heart rhythms and that it can be given concomitantly with other commonly used antiarrhythmic drugs," says Mehmet Aktas, M.D., associate professor of Cardiology and a member of the UR Medicine Heart & Vascular team. "This trial provides clinicians with one more treatment option for patients who have recurrent lethal arrhythmias, which is huge given that this is a very sick population for which we often have limited therapies."
Watch the video below to learn more about ventricular tachycardia (VT)!